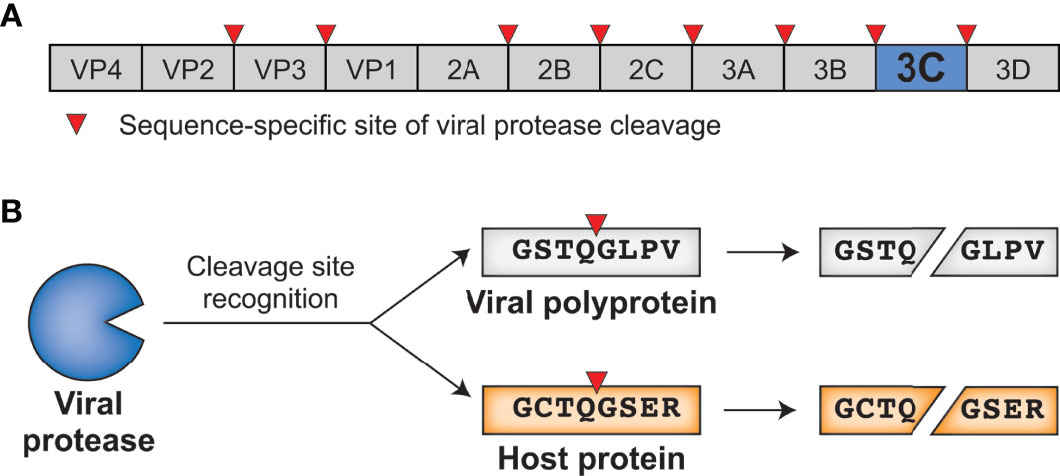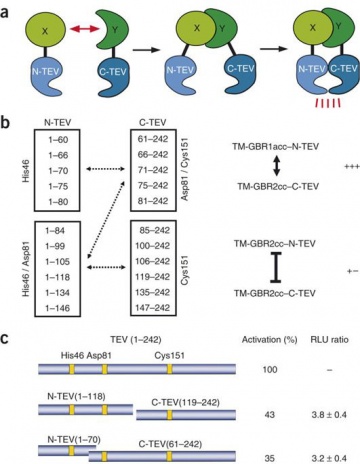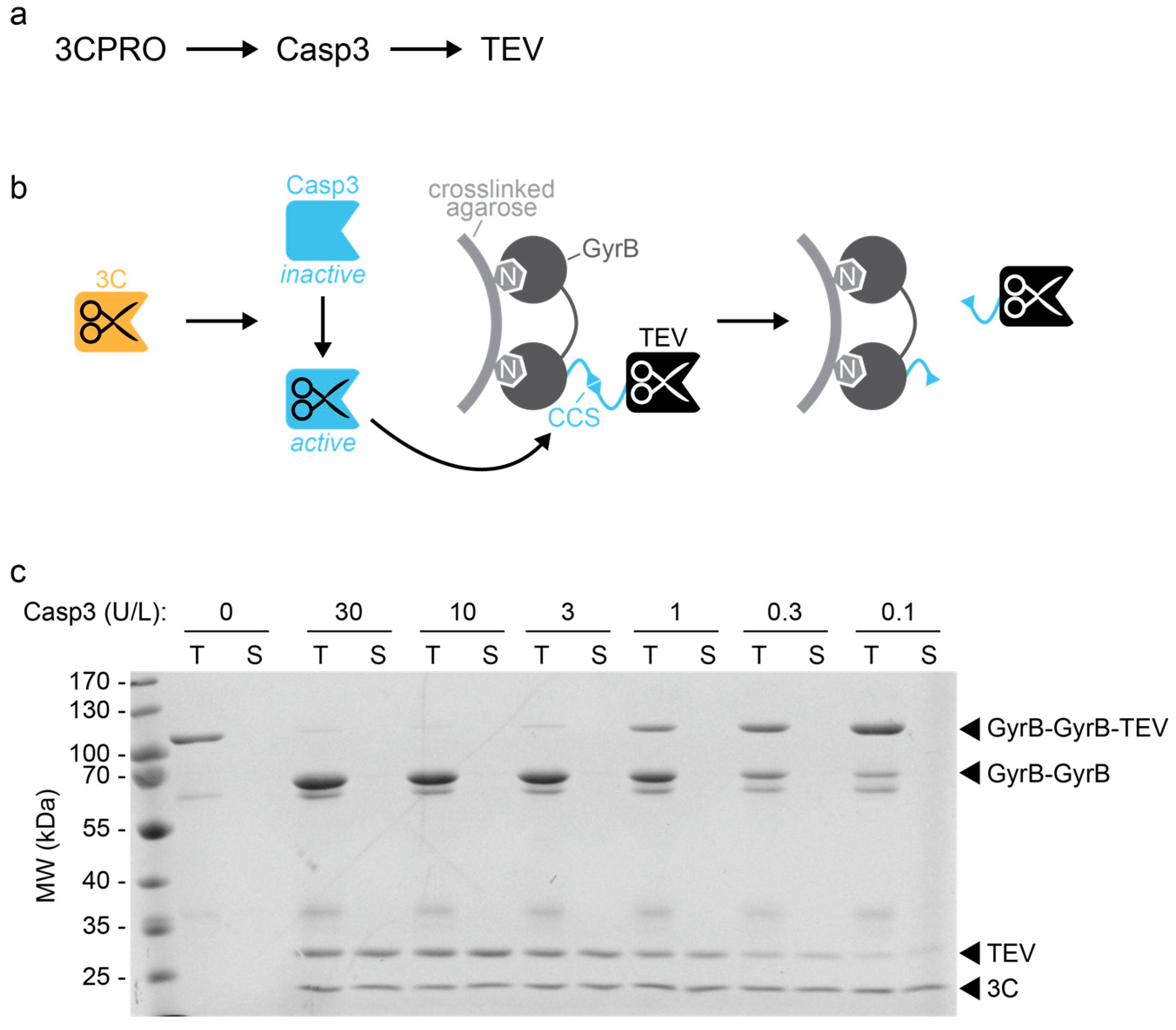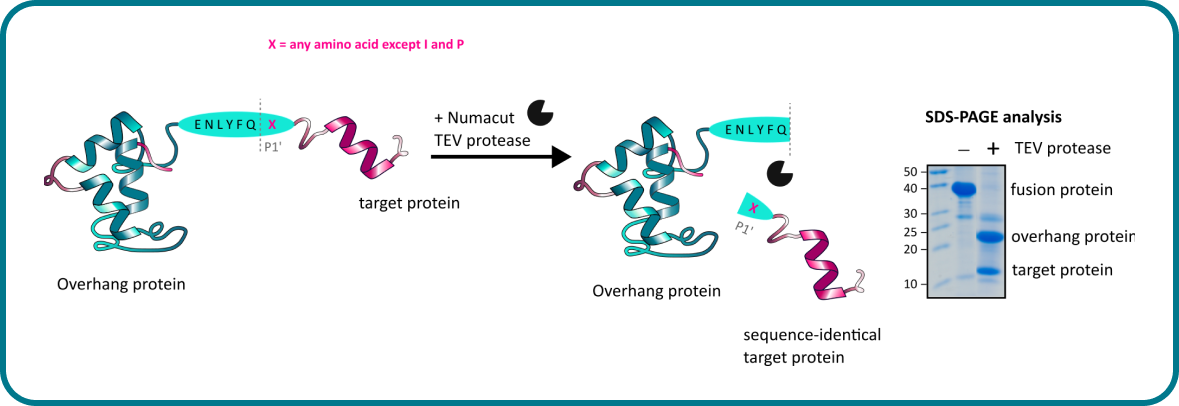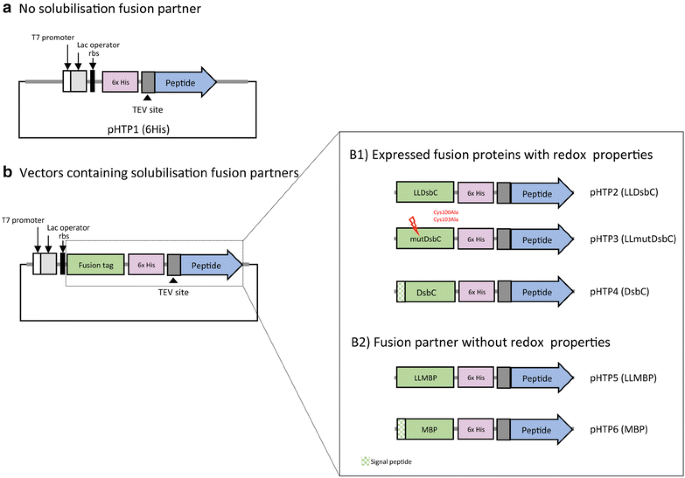
Gene design, fusion technology and TEV cleavage conditions influence the purification of oxidized disulphide-rich venom peptides in Escherichia coli | Microbial Cell Factories | Full Text

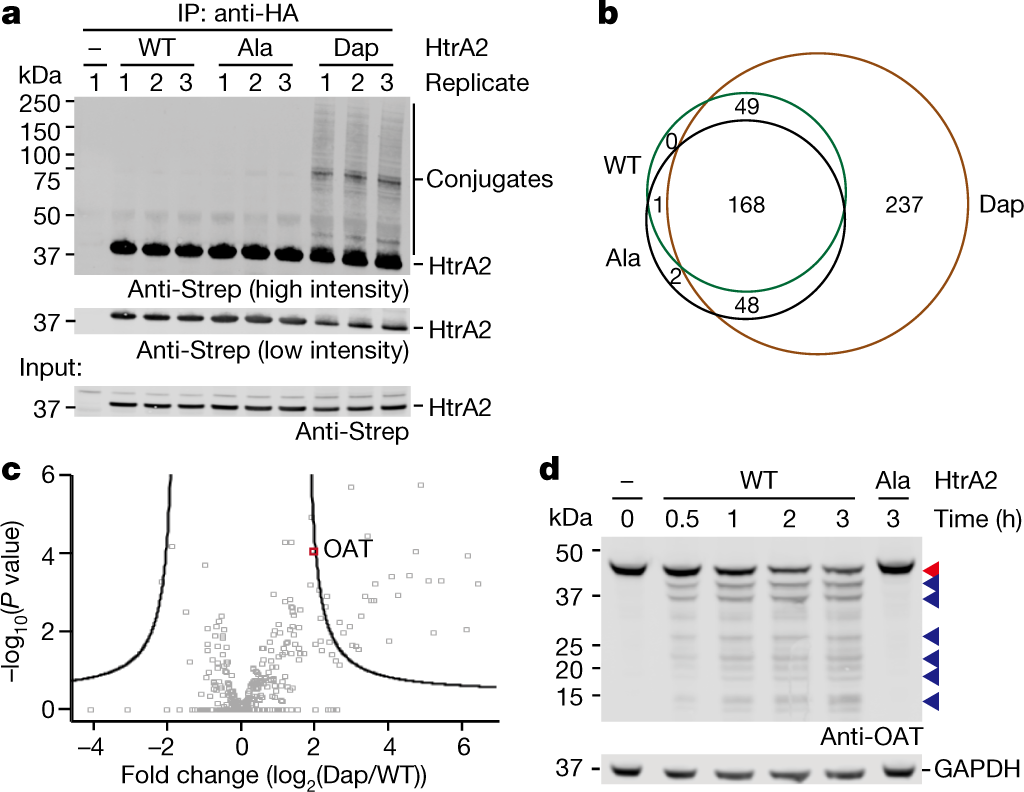
Deep profiling of protease substrate specificity enabled by dual random and scanned human proteome substrate phage libraries | PNAS

Schematic presentation of protease TEV cleavage site, V8 cleavage site... | Download Scientific Diagram
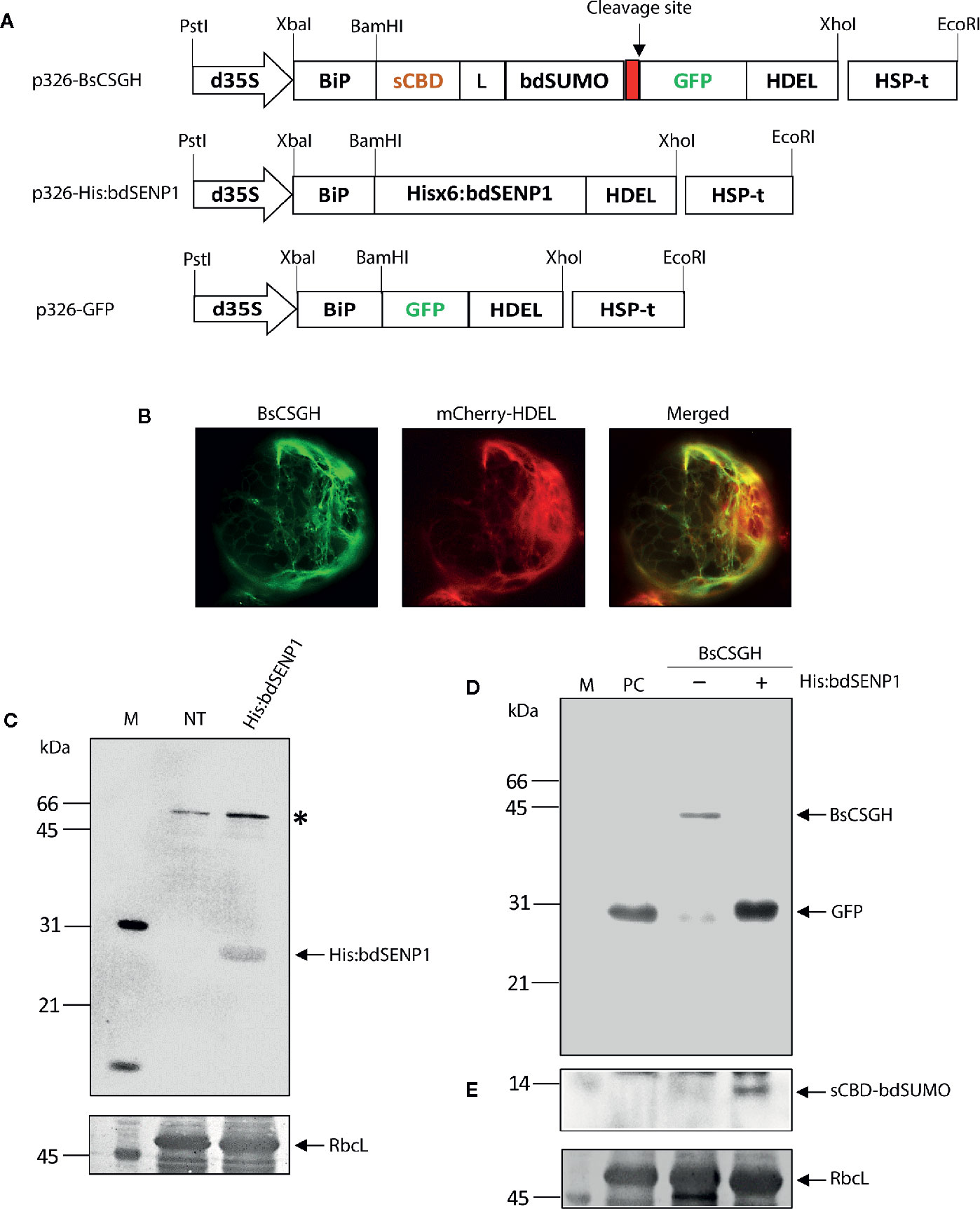
Frontiers | In Vivo Removal of N-Terminal Fusion Domains From Recombinant Target Proteins Produced in Nicotiana benthamiana

YESS 2.0, a Tunable Platform for Enzyme Evolution, Yields Highly Active TEV Protease Variants | ACS Synthetic Biology

Expression strategy of the CBD-lunasin fusion protein and subsequent... | Download Scientific Diagram

Going native: Complete removal of protein purification affinity tags by simple modification of existing tags and proteases - ScienceDirect

Global Sequencing of Proteolytic Cleavage Sites in Apoptosis by Specific Labeling of Protein N Termini: Cell

Three-Amino Acid Spacing of Presenilin Endoproteolysis Suggests a General Stepwise Cleavage of γ-Secretase-Mediated Intramembrane Proteolysis | Journal of Neuroscience

Three-Amino Acid Spacing of Presenilin Endoproteolysis Suggests a General Stepwise Cleavage of γ-Secretase-Mediated Intramembrane Proteolysis | Journal of Neuroscience

TEV protease cleavage of bioGATA-2 bound to streptavidin beads. (A)... | Download Scientific Diagram
Highly efficient soluble expression, purification and characterization of recombinant Aβ42 from Escherichia coli
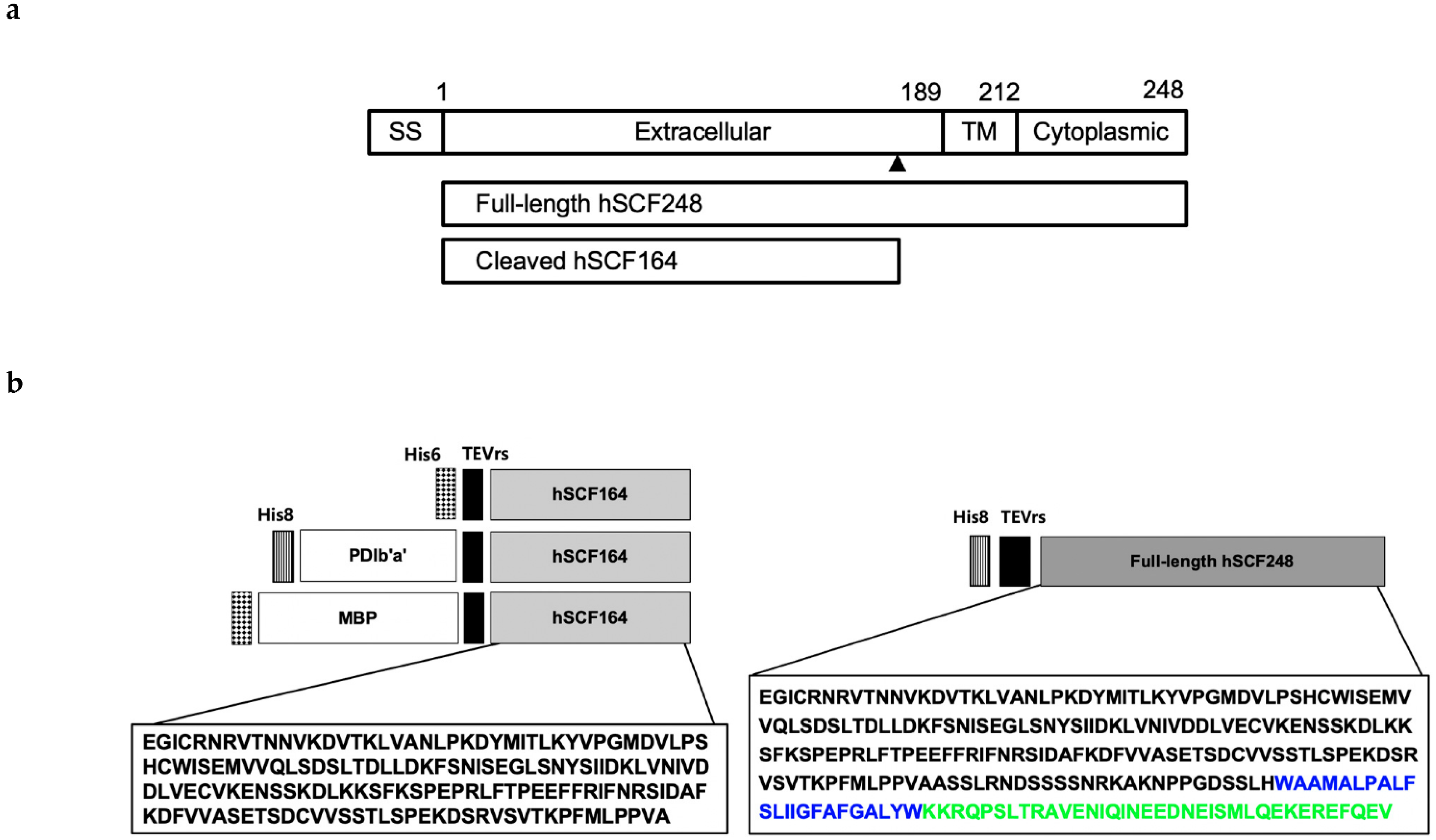
IJMS | Free Full-Text | Novel Bacterial Production of Two Different Bioactive Forms of Human Stem-Cell Factor

Improved yield, stability, and cleavage reaction of a novel tobacco etch virus protease mutant | SpringerLink