Production of non‐glycosylated recombinant proteins in Nicotiana benthamiana plants by co‐expressing bacterial PNGase F - Mamedov - 2012 - Plant Biotechnology Journal - Wiley Online Library

A Novel PNGase Rc for Improved Protein N-Deglycosylation in Bioanalytics and Hydrogen–Deuterium Exchange Coupled With Mass Spectrometry Epitope Mapping under Challenging Conditions | Analytical Chemistry

Site-specific, covalent immobilization of PNGase F on magnetic particles mediated by microbial transglutaminase - ScienceDirect
Enhanced Recombinant Protein Production of Soluble, Highly Active and Immobilizable PNGase F | SpringerLink

Figure 3 | Furin Functions as a Nonproteolytic Chaperone for Matrix Metalloproteinase-28: MMP-28 Propeptide Sequence Requirement
Cloning and expression of peptide-N4-(N-acetyl-beta-D-glucosaminyl)asparagine amidase F in Escherichia coli.

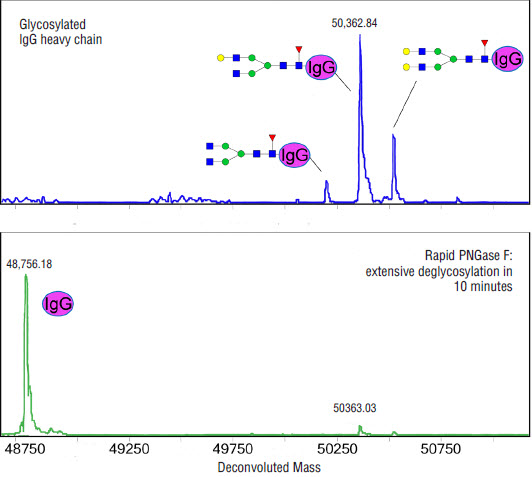
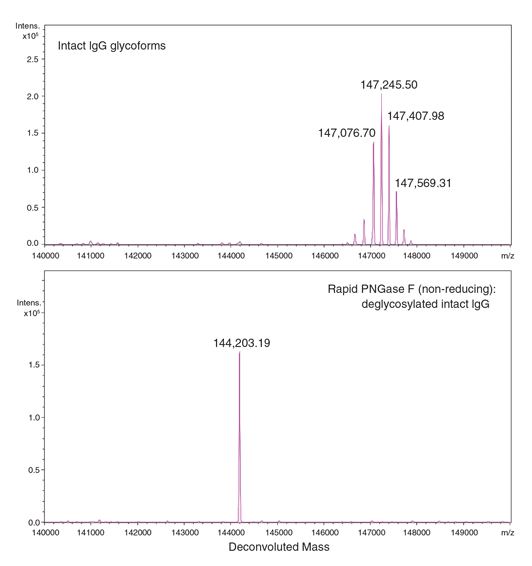
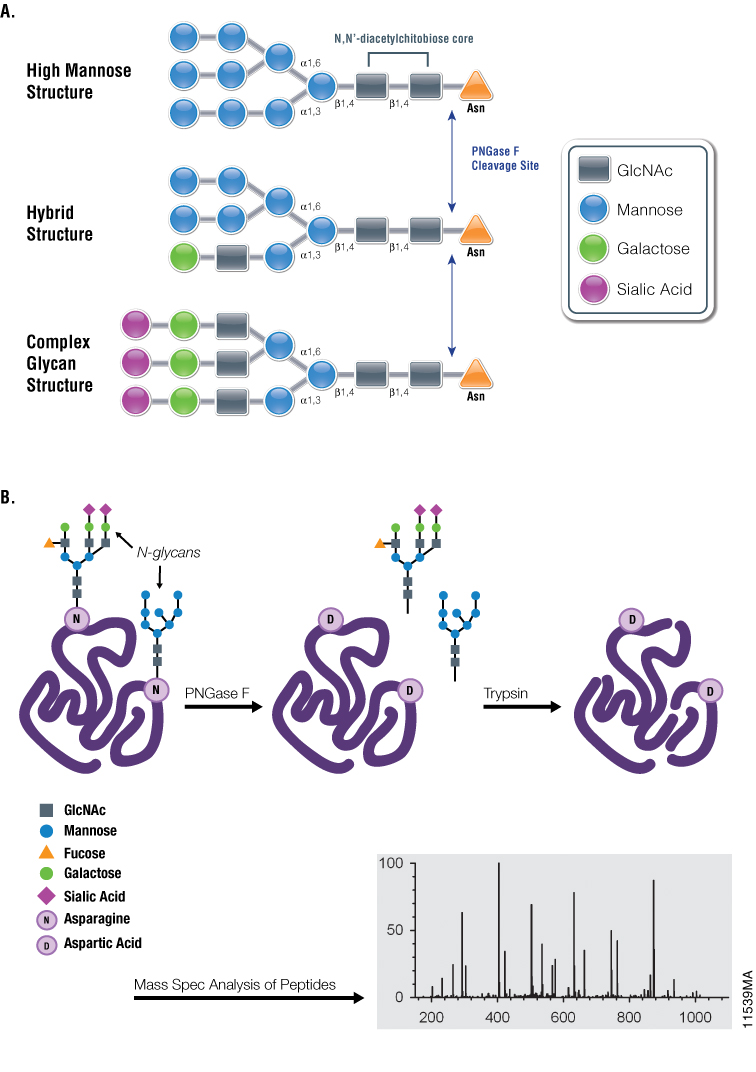
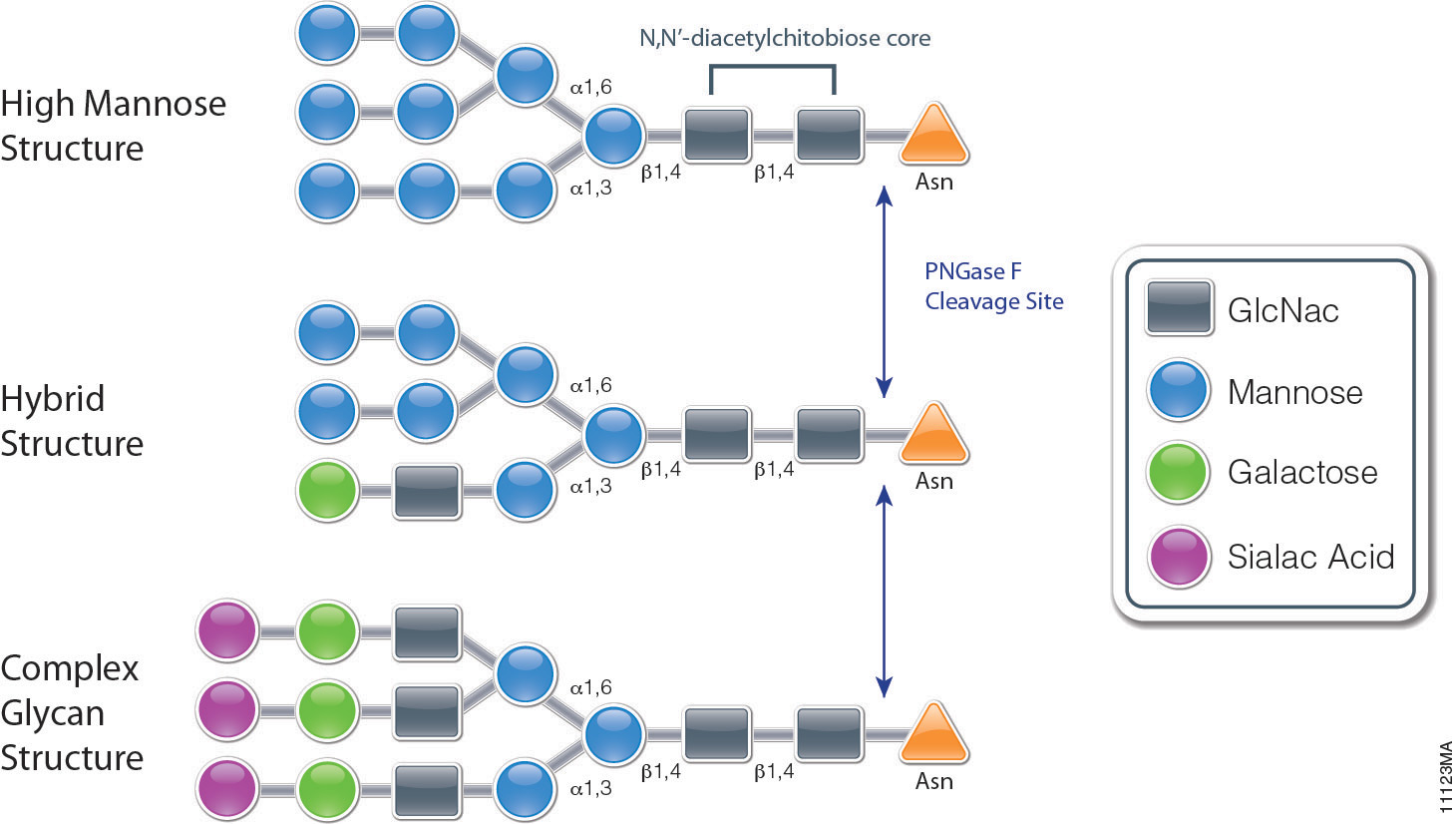
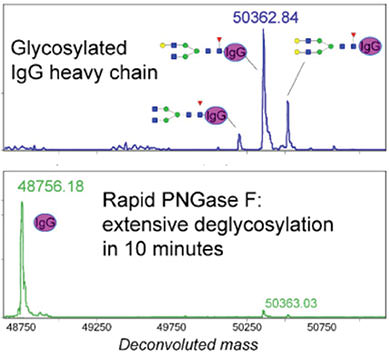
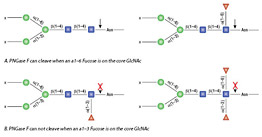
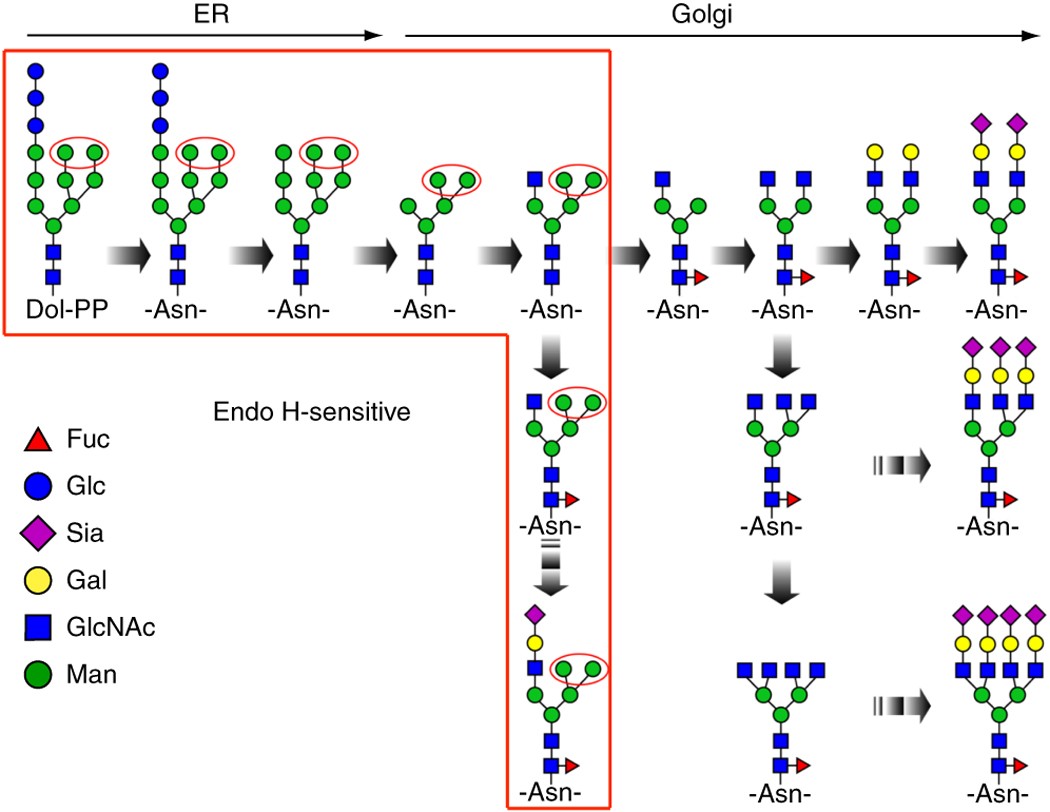
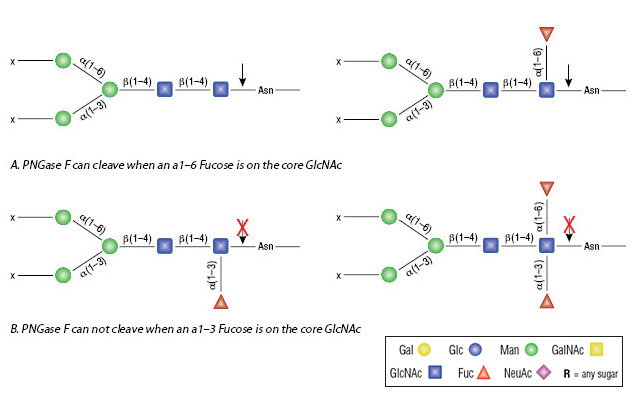
Using Glycosidases to Remove, Trim, or Modify Glycans on Therapeutic Proteins - BioProcess InternationalBioProcess International