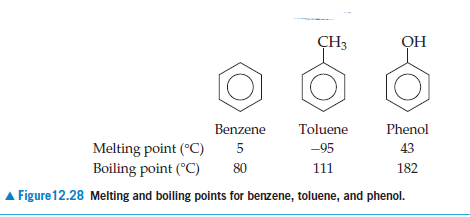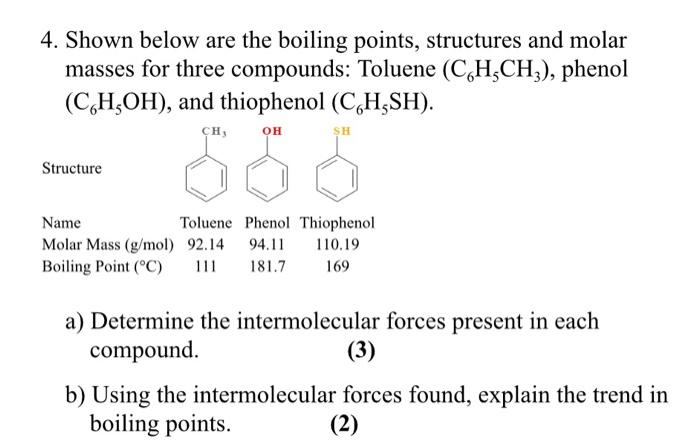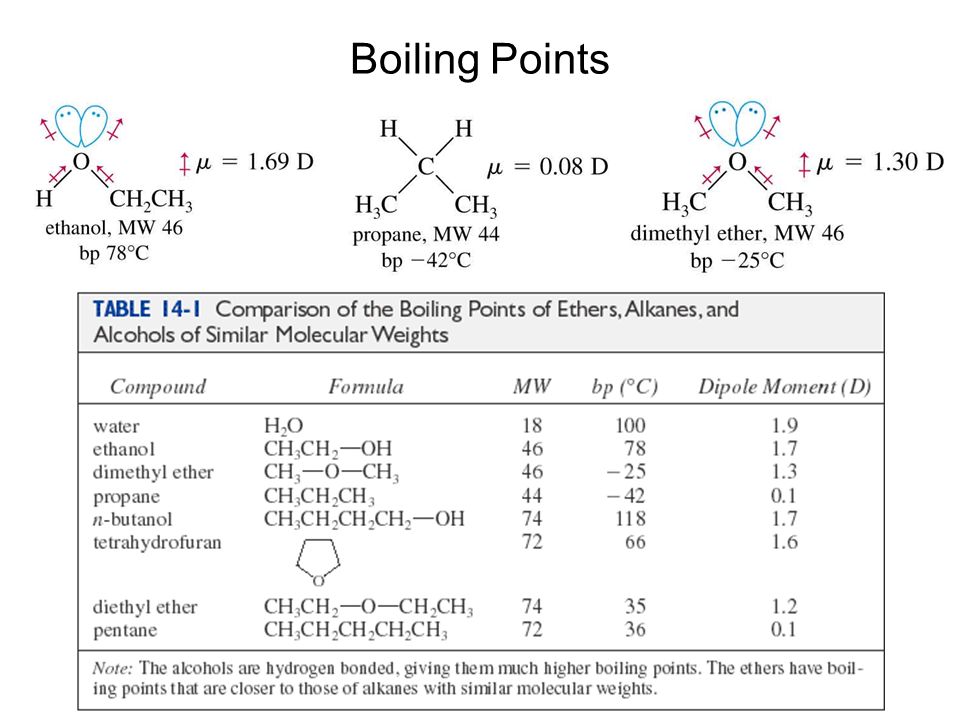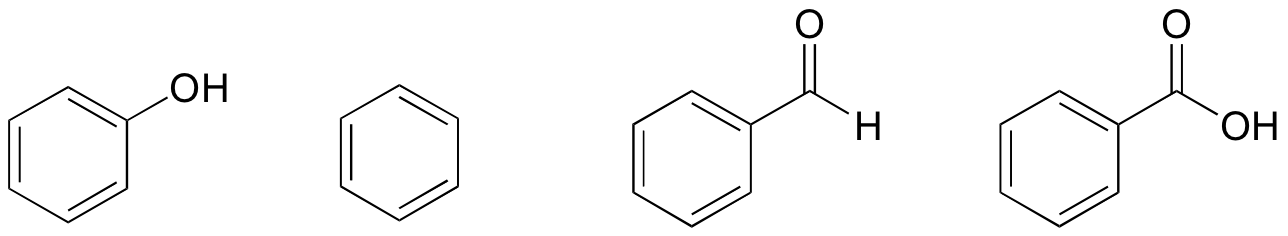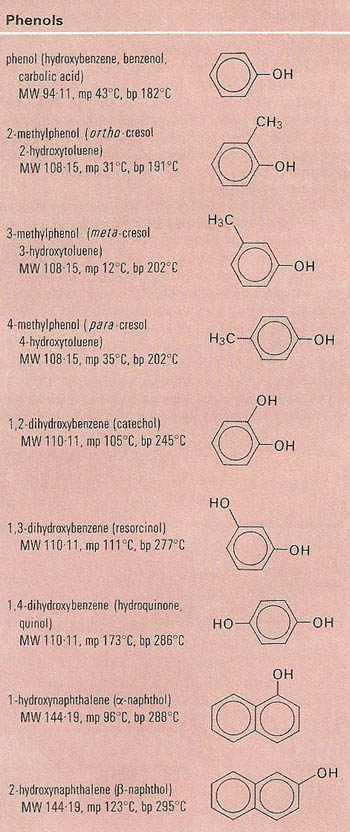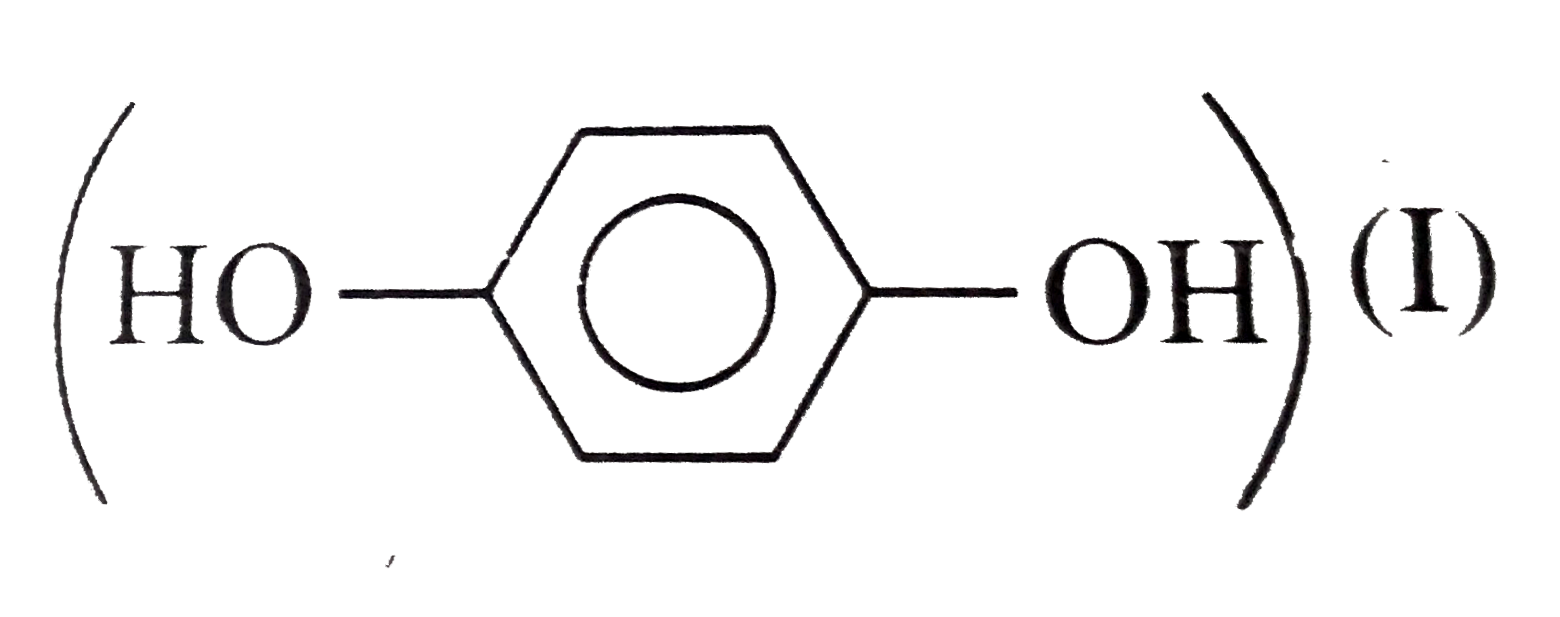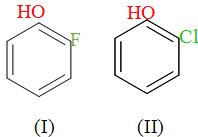
The correct order of boiling point is:\n \n \n \n \n \n \n \n \n \n (a) I II III(b) III II I(c) II I III(d) I III II
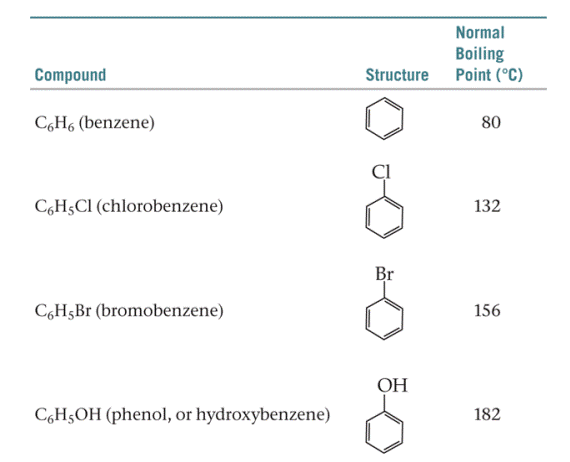
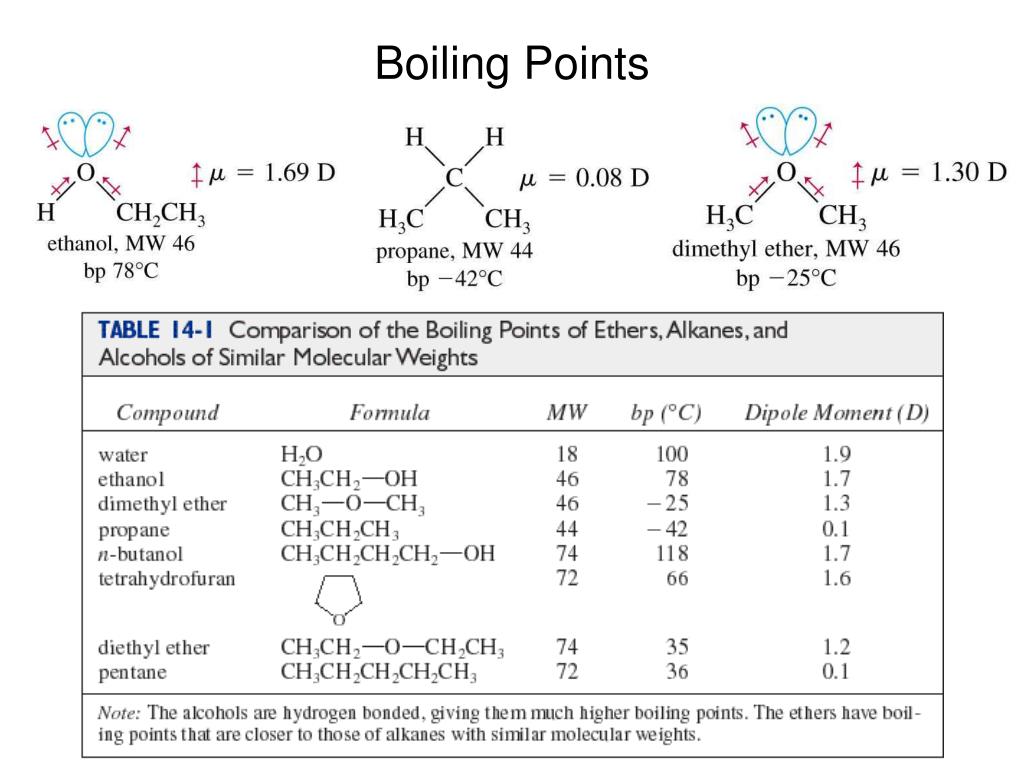
The table below shows the normal boiling points of benzene and benzene derivatives. How many of these compounds exhibit dispersion interaction? How many of these compounds exhibit dipole-dipole interaction? How many of
What is the increasing order of boiling point and solubility in water of alcohol and phenol? Give reasons also.
Why is the boiling point of phenol greater than alcohol? They both are containing the OH group. - Quora