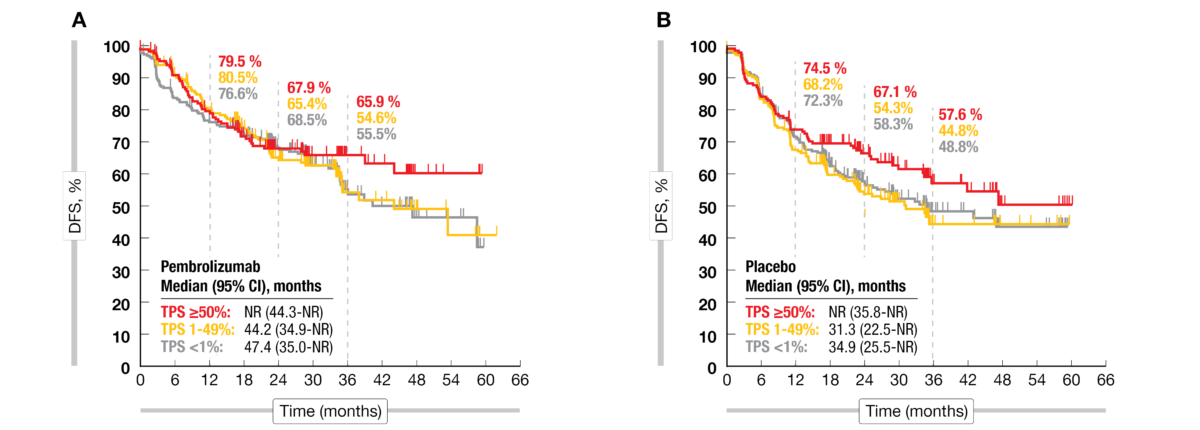
Stephen V Liu, MD on Twitter: "The #ESMOVirtualPlenary presented by Dr. @LuisPaz_Ares presents results from EORTC-1416-LCG / ETOP 8-15 / PEARLS / KEYNOTE-091: adjuvant pembrolizumab versus placebo for resected early stage #NSCLC. @

Mary O'Brien, MD @drMaryOBr @royalmarsdenNHS #ASCO22 #OncoTwitter Phase III PEARLS/KEYNOTE-091 Discussion

Gaining an Advantage Over NSCLC: How to Achieve the Greatest Benefit With Immunotherapy From Advanced to

Stephen V Liu, MD on Twitter: "#ASCO22 While PEARLS did meet its endpoint, we already have atezolizumab approved in this space after IMpower 010. Notes differences between these trials and again -

Pembrolizumab versus placebo as adjuvant therapy for completely resected stage IB–IIIA non-small-cell lung cancer (PEARLS/KEYNOTE-091): an interim analysis of a randomised, triple-blind, phase 3 trial - The Lancet Oncology

KEYTRUDA® (pembrolizumab) Showed Statistically Significant Improvement In Disease-Free Survival Versus Placebo As Adjuvant Treatment For Patients With Stage IB-IIIA Non-Small Cell Lung Cancer Regardless Of PD-L1 Expression 2023 - EORTC
Pembrolizumab versus placebo as adjuvant therapy for completely resected stage IB–IIIA non-small-cell lung cancer (PEARLS/KEYNOTE-091): an interim analysis of a randomised, triple-blind, phase 3 trial | AIOM

Aɴᴛᴏɴɪᴏ Pᴀssᴀʀᴏ on Twitter: "🚨 Adjuvant pembrolizumab for early-stage NSCLC following complete resection: the ph III PEARLS(KEYNOTE-091) by Luis Paz Ares @ETOP_eu @EORTC #LCSM Primary endpoint: DSF in overall population and PDL1

Stephen V Liu, MD on Twitter: "#ASCO22 Discussion of PEARLS (KEYNOTE-091, @ETOP_eu 8-15), a phase III study of adjuvant pembrolizumab in resected NSCLC. The study did meet its primary endpoint (DFS HR