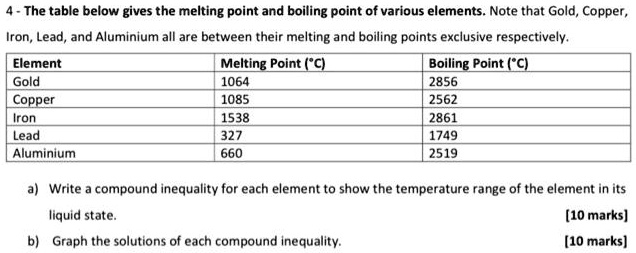
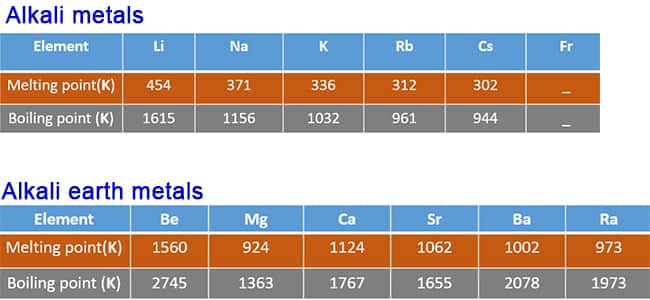
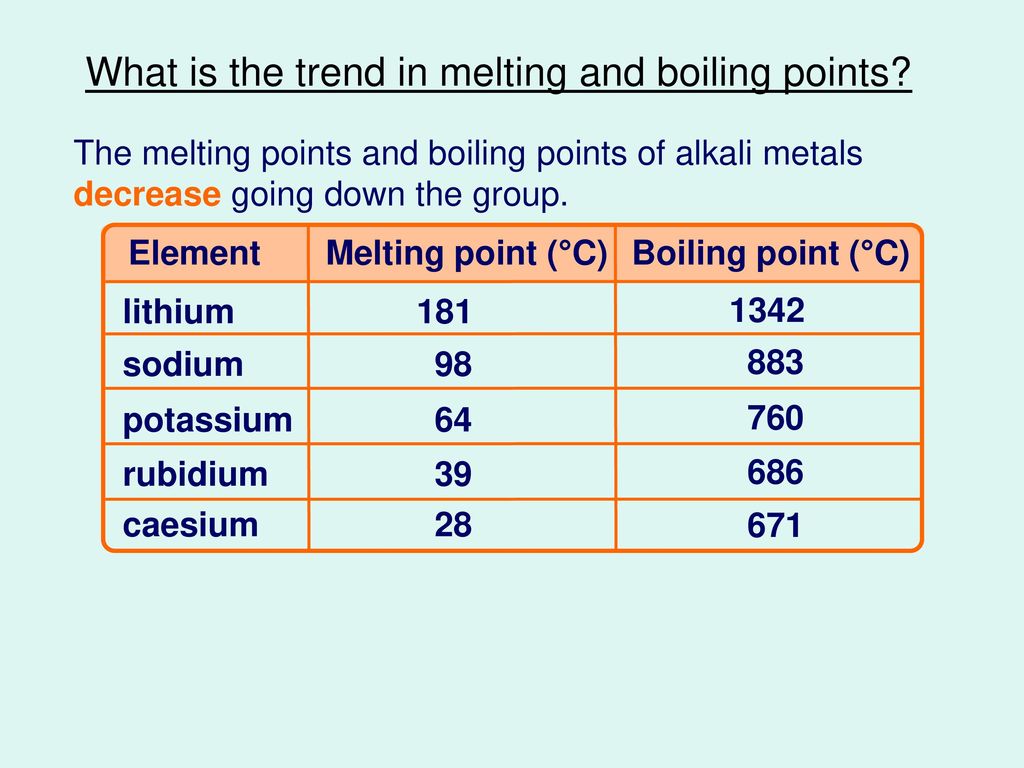
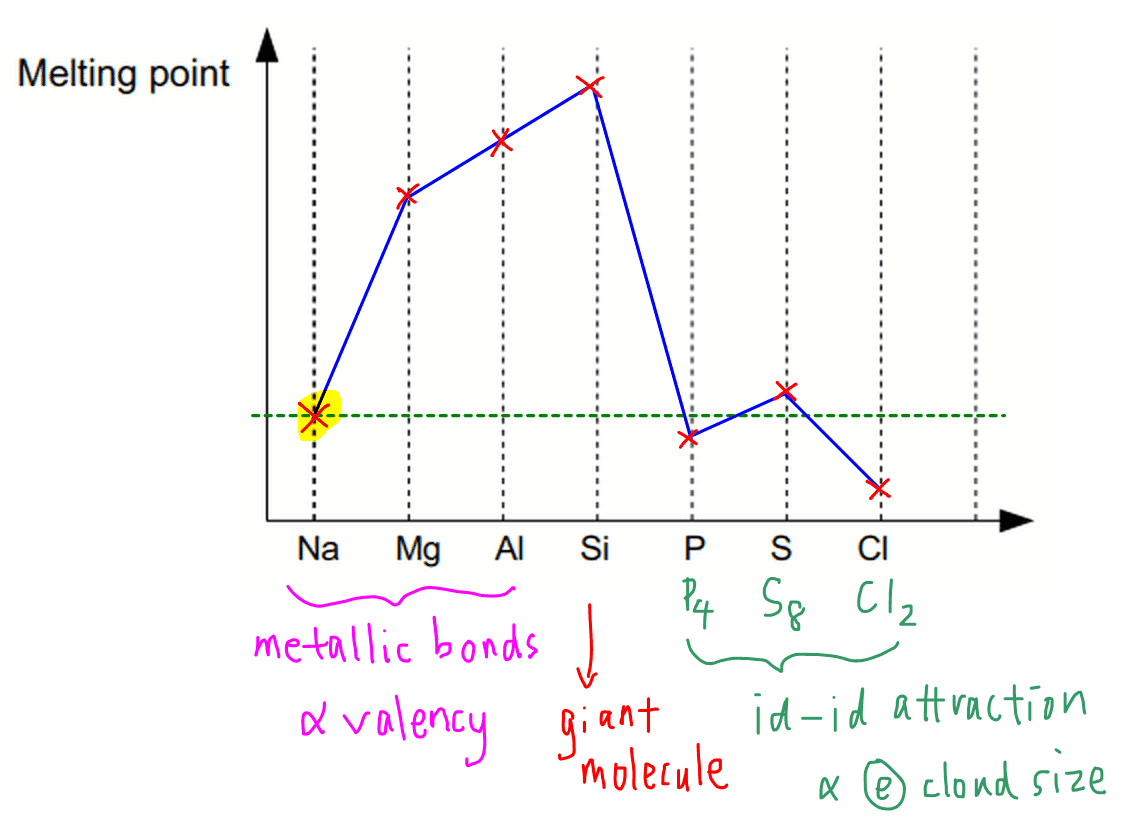
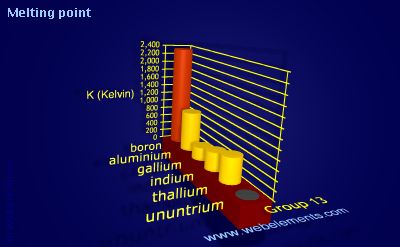
A Level Period 2 trends and explanations of physical properties of lithium beryllium boron carbon nitrogen oxygen fluorine neon periodic table GCE AS A2 inorganic revision notes KS5

A Level Z = 1 to 20 Periodicity plots - graphs of physical properties of elements of the Periodic Table GCE AS A2 inorganic revision notes KS5

thermodynamics - Why do mercury, cadmium and zinc have low melting and boiling points and elements next to them start melting at a bit higher temperatures? - Physics Stack Exchange

Elements General Physical Properties : Atomic Size | Melting point | Boiling point - The Chemistry Guru

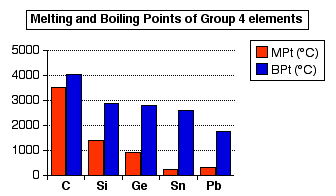
Why do the melting point and boiling point of transition metals high? | by KAKALI GHOSH , Teacher,blogger. M.Sc chemistry. | Medium
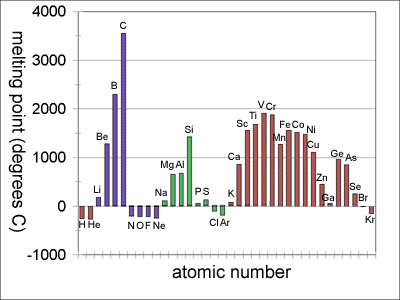
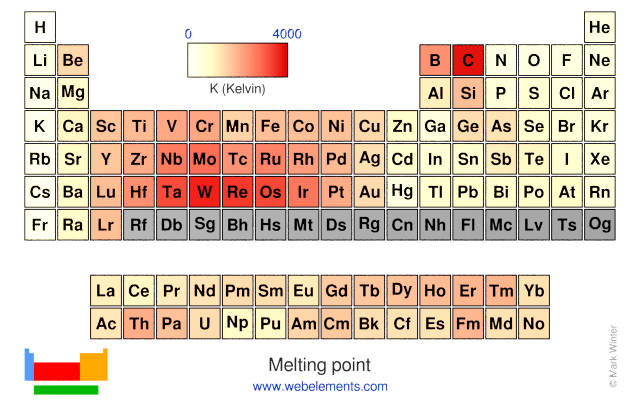
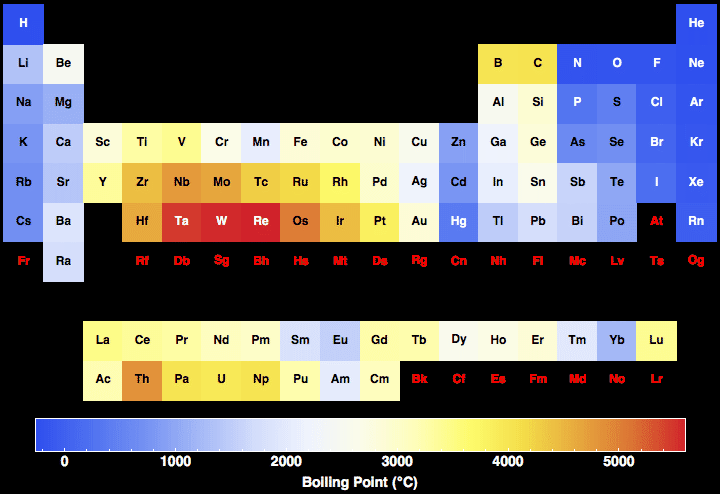
General Chemistry Online: FAQ: The periodic table: Is there a trend in melting points on the periodic table?