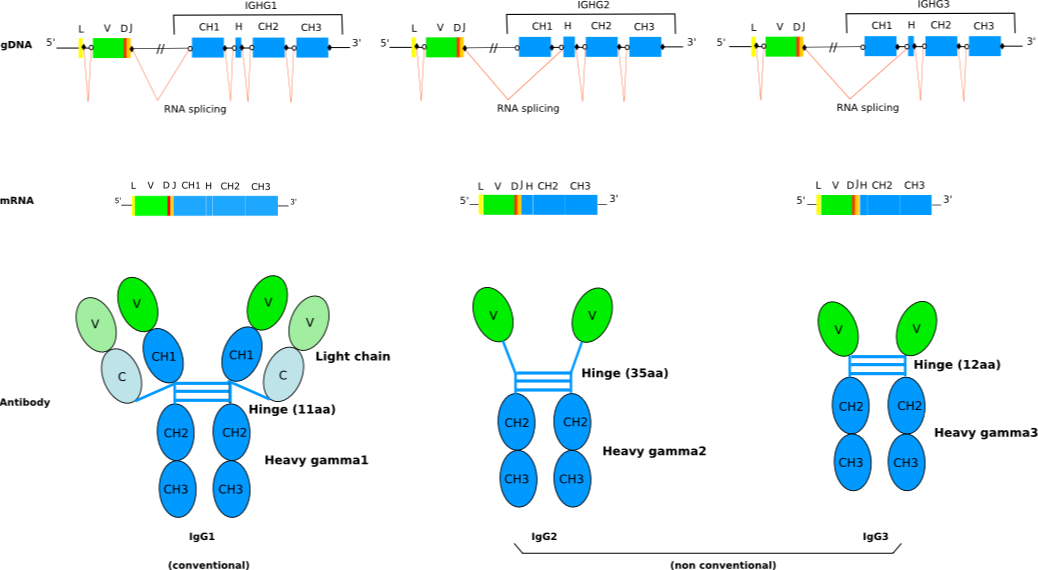
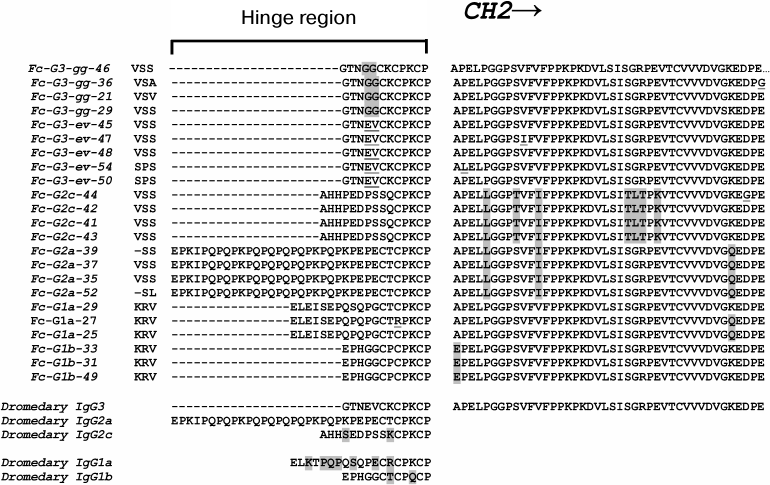
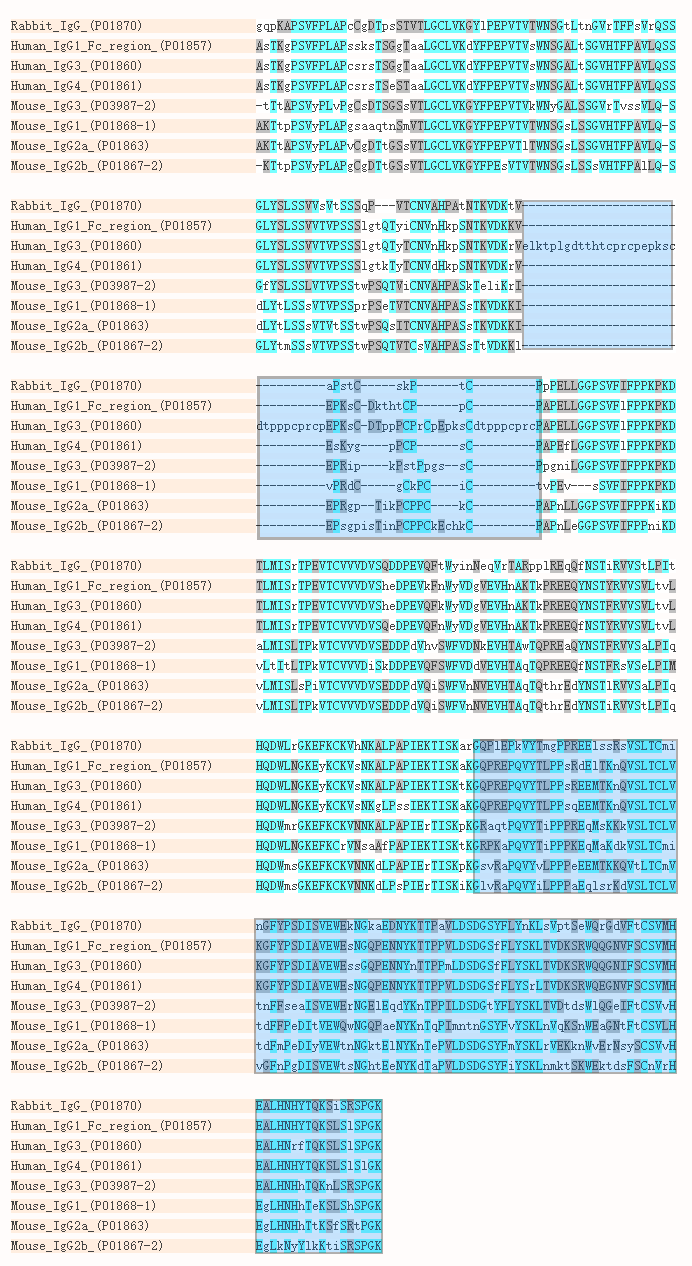
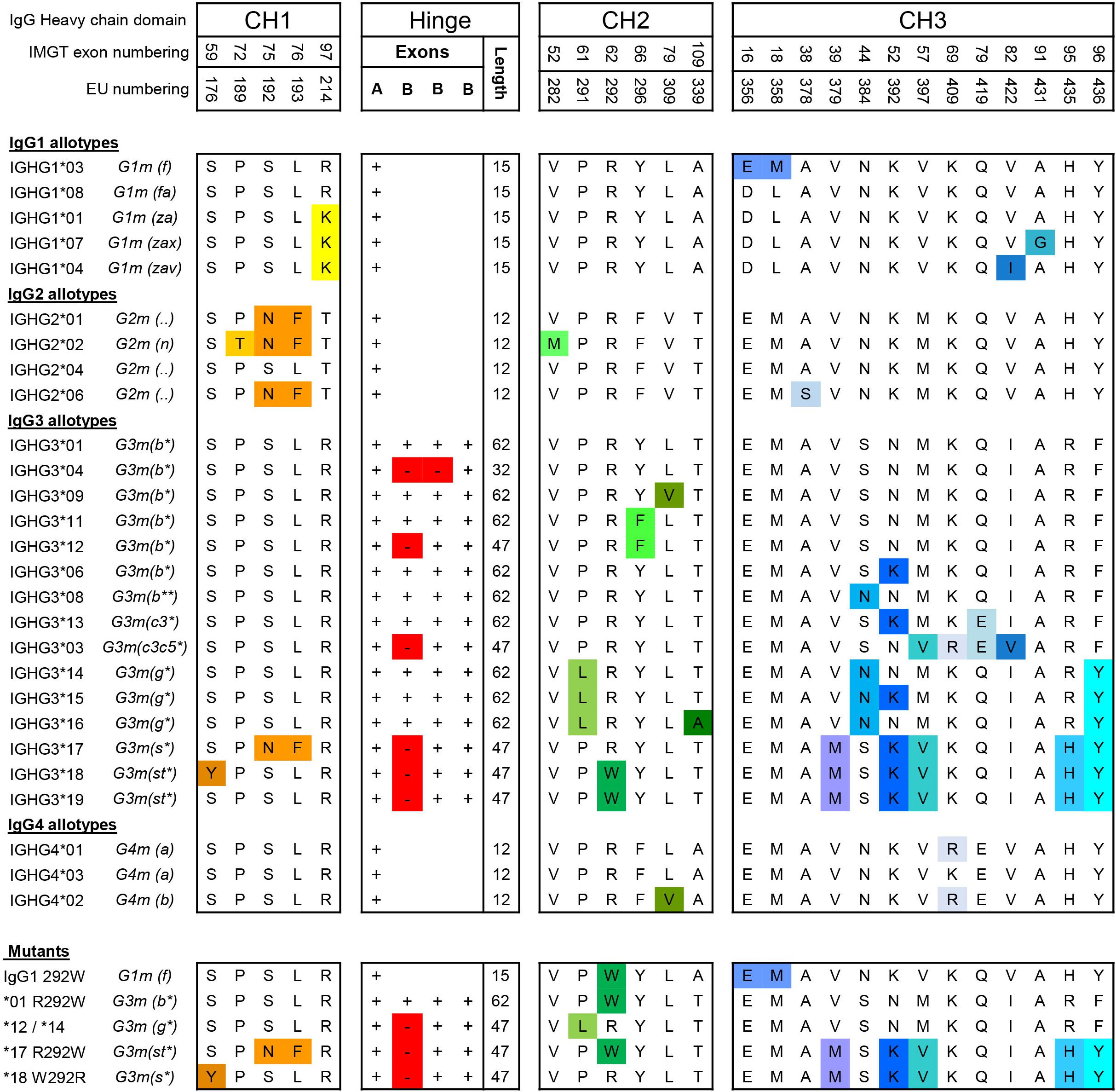
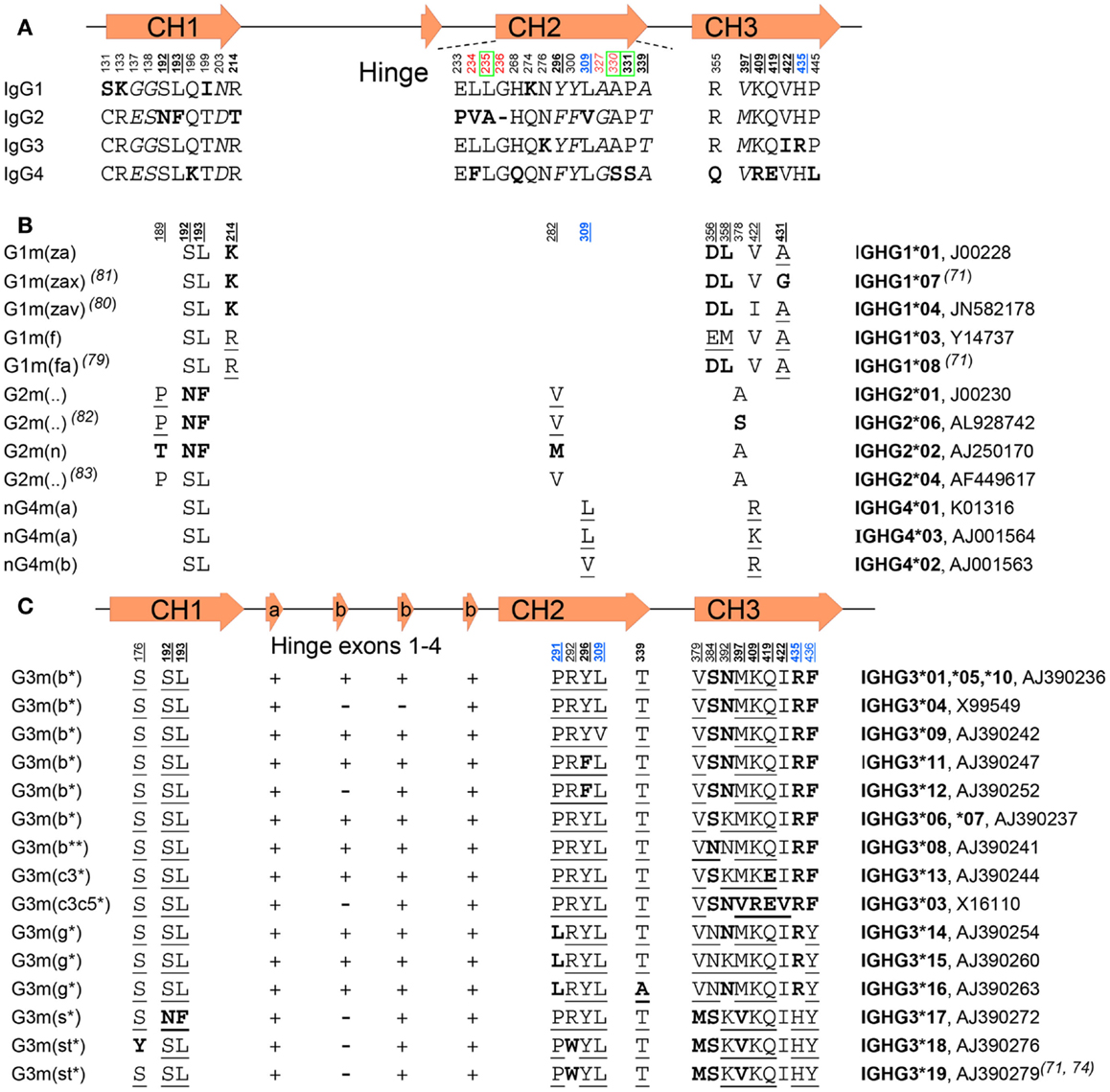
Engineering the hinge region of human IgG1 Fc-fused bispecific antibodies to improve fragmentation resistance | Scientific Reports

Predicting amino acid sequences of the antibody human VH chains from its first several residues | PNAS

Amino acid sequence alignment of the Fc region of eqIgGs. The sequence... | Download Scientific Diagram

Figure 1 from Functional, Biophysical, and Structural Characterization of Human IgG1 and IgG4 Fc Variants with Ablated Immune Functionality | Semantic Scholar
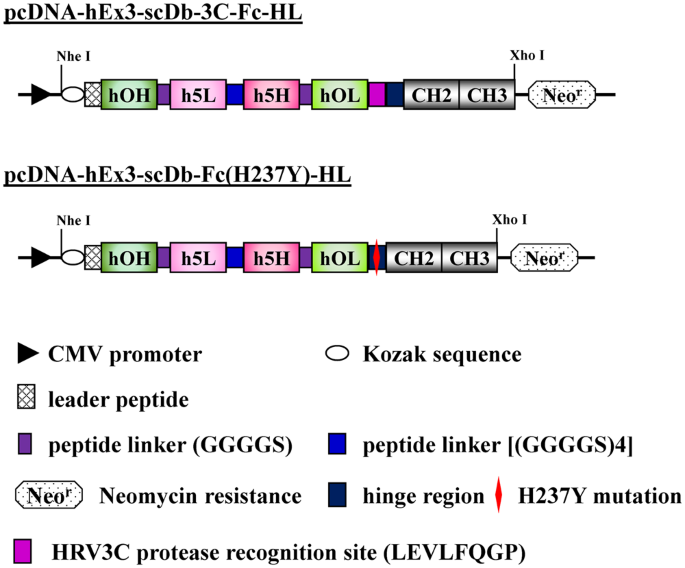
Engineering the hinge region of human IgG1 Fc-fused bispecific antibodies to improve fragmentation resistance | Scientific Reports
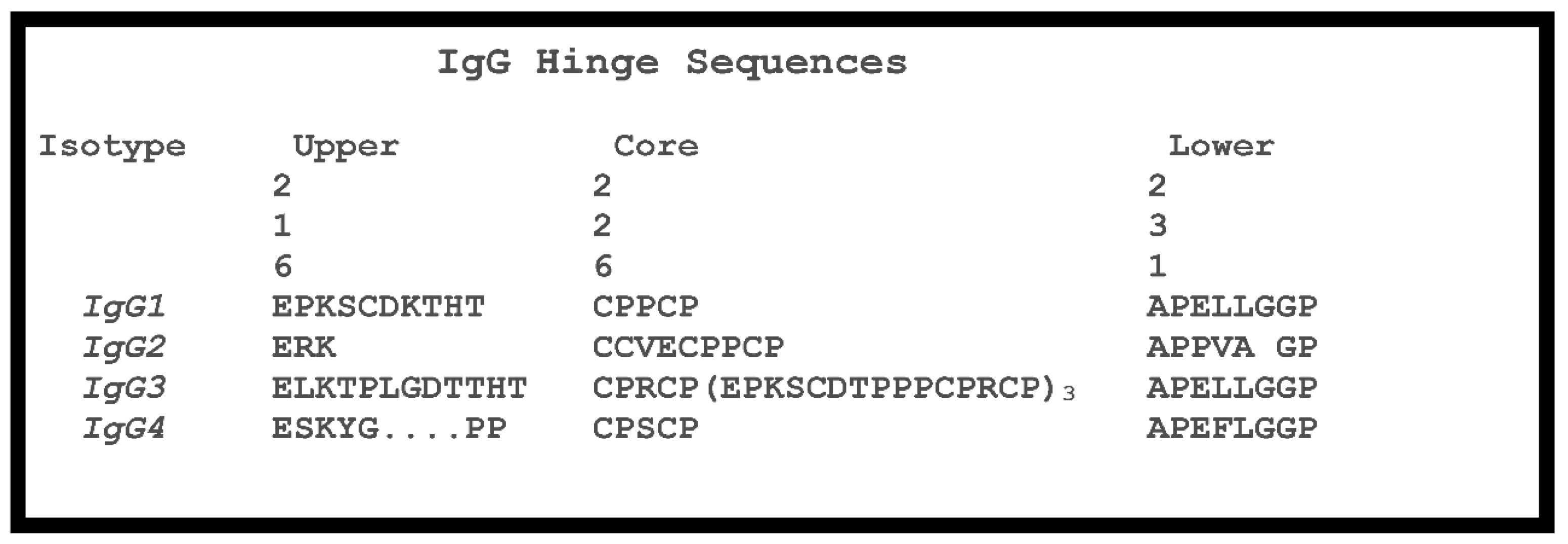
Antibodies | Free Full-Text | Antibody Structure and Function: The Basis for Engineering Therapeutics

Pharmacokinetic Properties of Humanized IgG1 and IgG4 Antibodies in Preclinical Species: Translational Evaluation | SpringerLink

Molecular sequence of human and cynomolgus IgG and FcgR. (A) Amino acid... | Download Scientific Diagram

Identification of dentinogenic cell-specific surface antigens in odontoblast-like cells derived from adult dental pulp | Stem Cell Research & Therapy | Full Text

Production of IgG1-based bispecific antibody without extra cysteine residue via intein-mediated protein trans-splicing | Scientific Reports
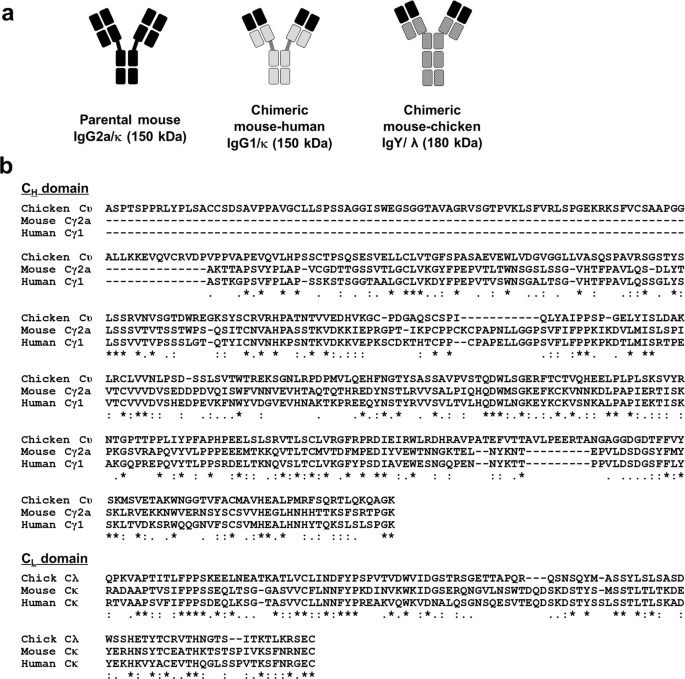
Antigen-binding affinity and thermostability of chimeric mouse-chicken IgY and mouse-human IgG antibodies with identical variable domains | Scientific Reports

Figure 1 from The Origins, Specificity, and Potential Biological Relevance of Human Anti-IgG Hinge Autoantibodies | Semantic Scholar