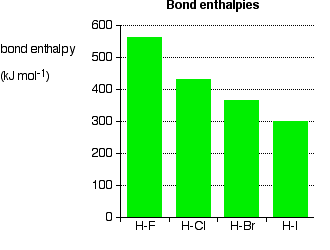
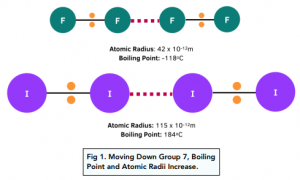
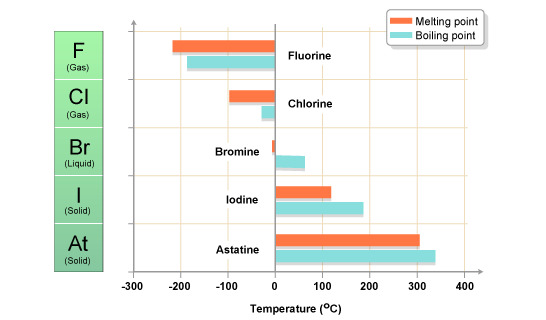
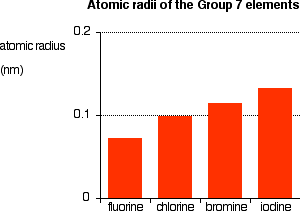
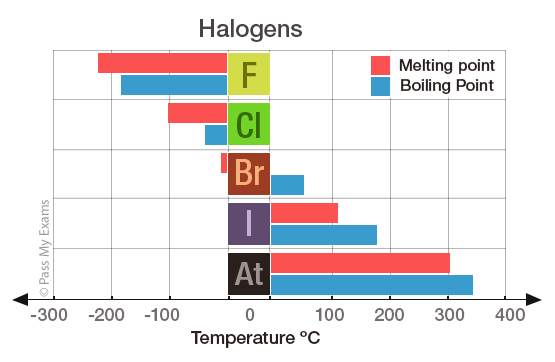
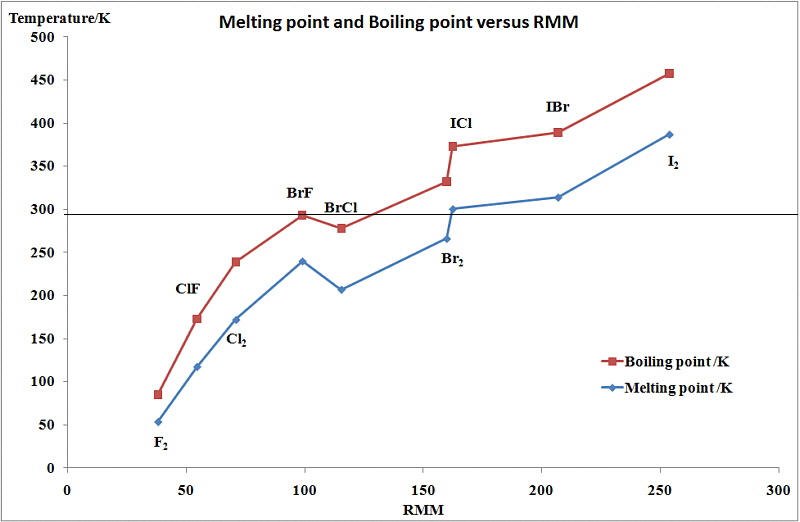
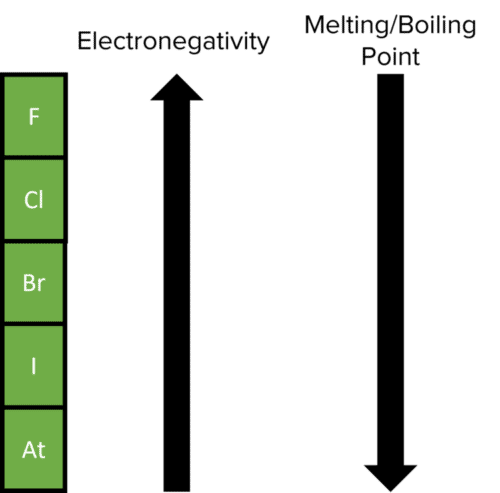
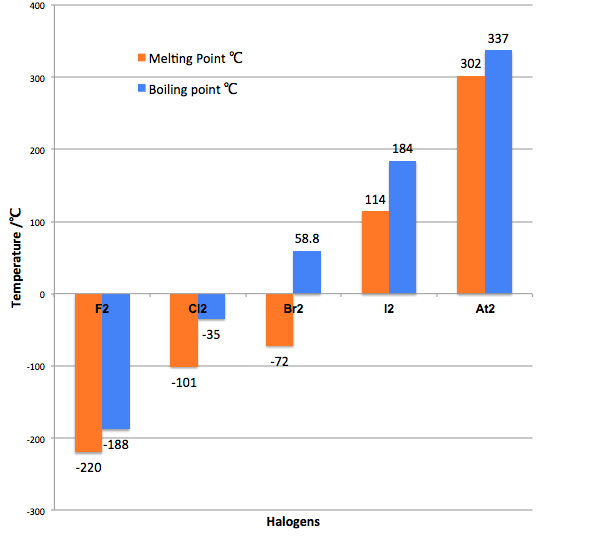
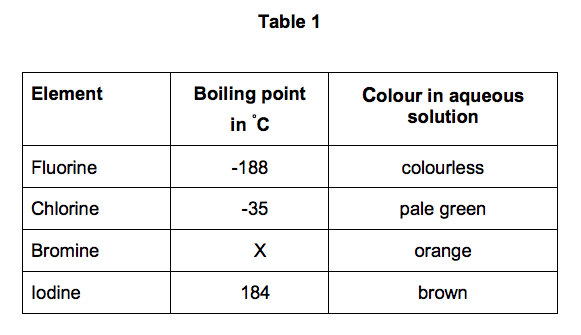
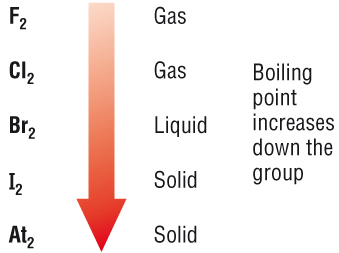
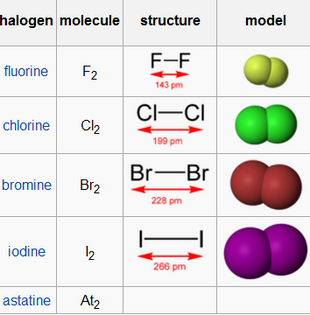
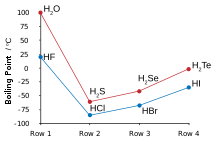
Group 7 HALOGENS fluorine chlorine bromine iodine physical properties balanced equations chemical reactions balanced gcse chemistry revision notes KS4 science igcse O level

Which of the following statements best explains the trends in boiling points? A.)The atomic size increases - Brainly.com

The table below shows the boiling points of some halogens. Explain the trend in the boiling points of - Brainly.com