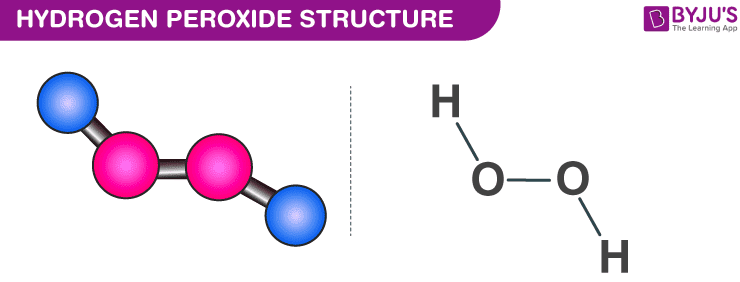
Molecular solids generally have low melting points and boiling points due to relatively weak intermolecular attractions that hold the molecules in their solid form.Molecular SolidBoiling Point (^∘C) H2Te 0 H2Se - 50

SOLVED: The temperature at which the following process reaches equilibrium at 1.0 atm is the normal boiling point of hydrogen peroxide: H20z(0) = HzOzlg) Use the 298 K thermodynamic information given below

Which of the following is not a proper match(A) D-D is equal to H-H........ Bond length(B) H20 Ꭰ20 , H2O2 - Brainly.in

Effect of temperature and H2O2 concentration on the carboxyl groups'... | Download Scientific Diagram



.gif)