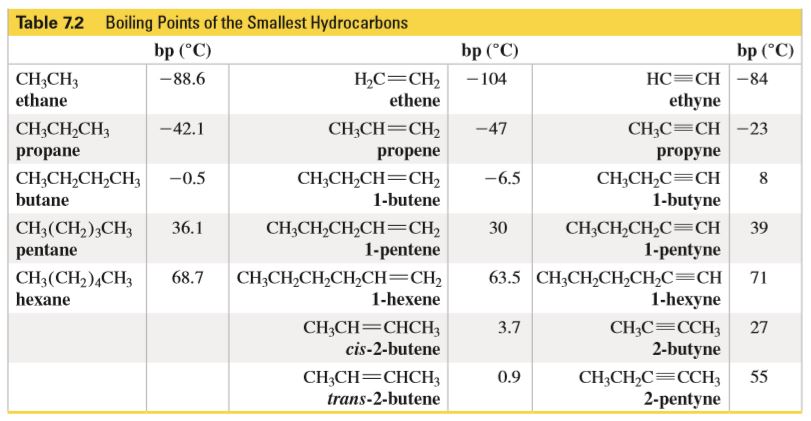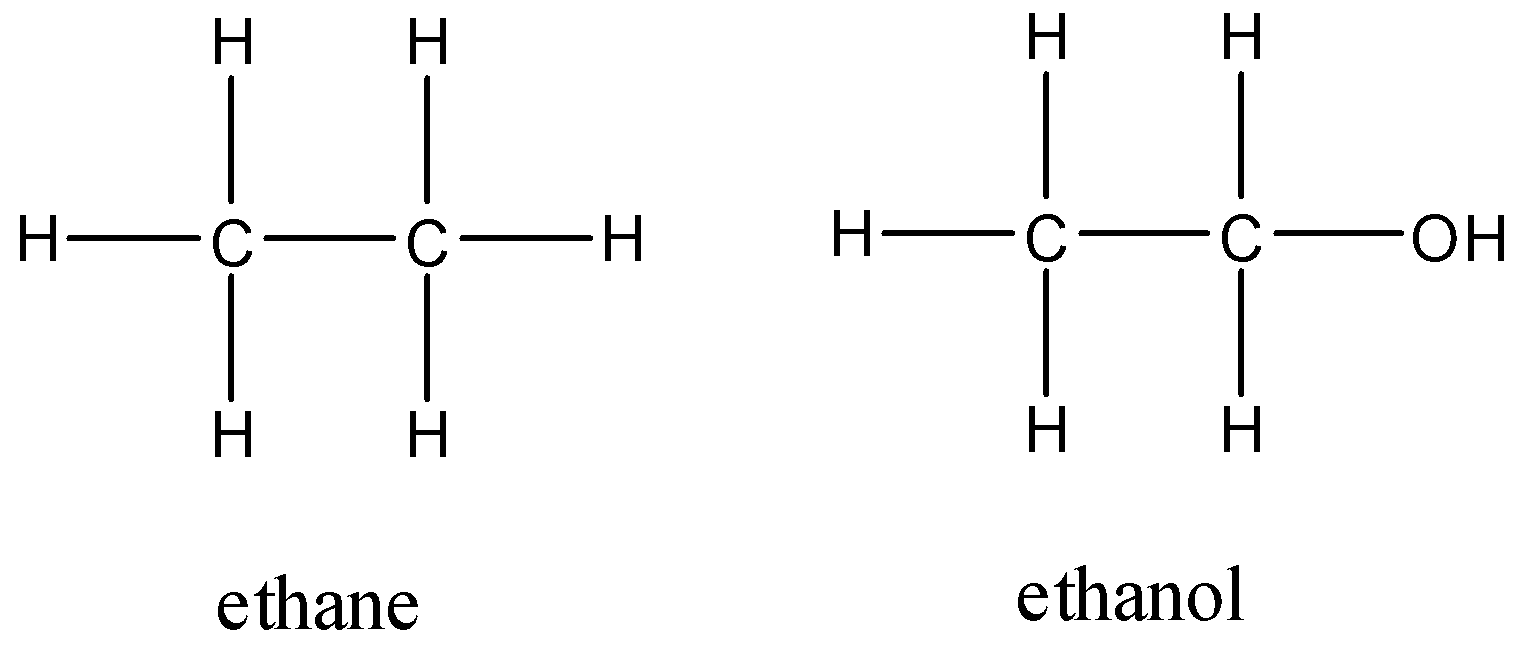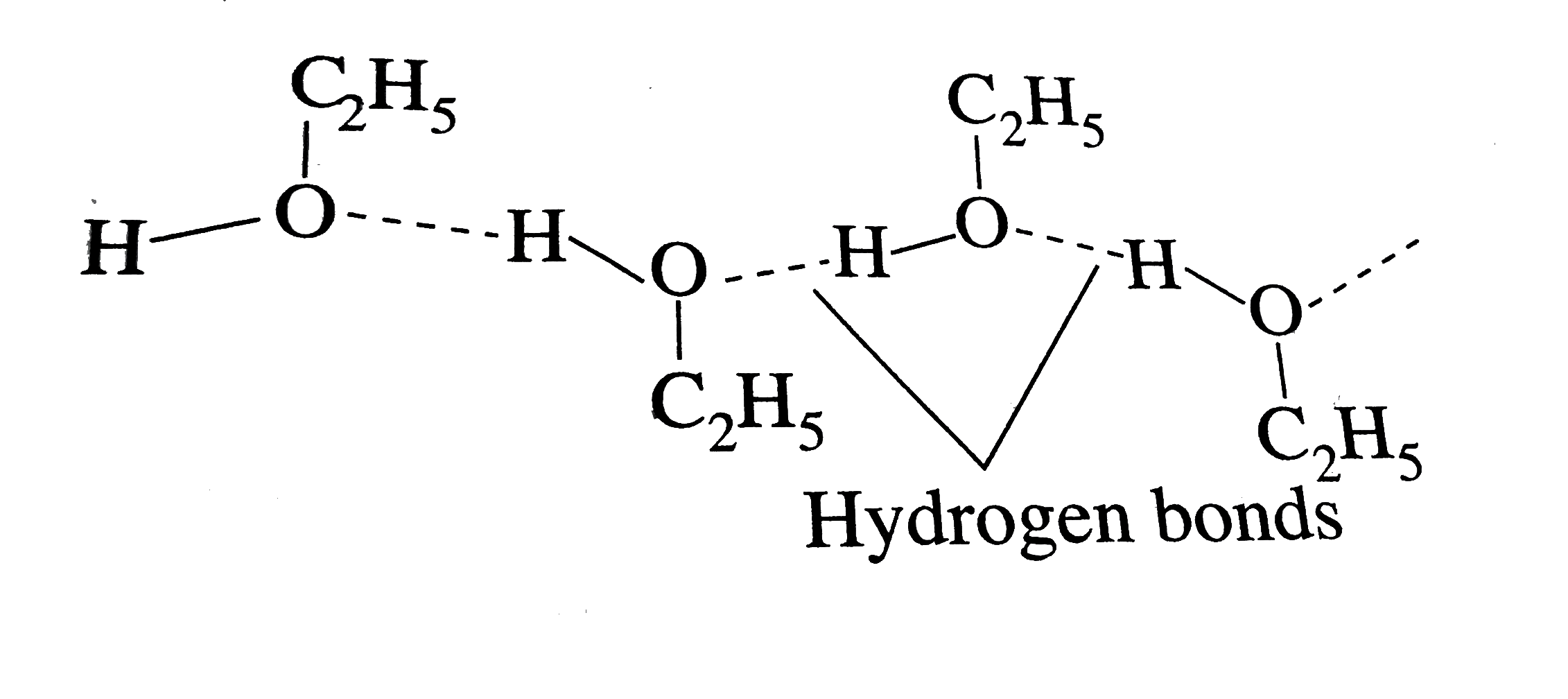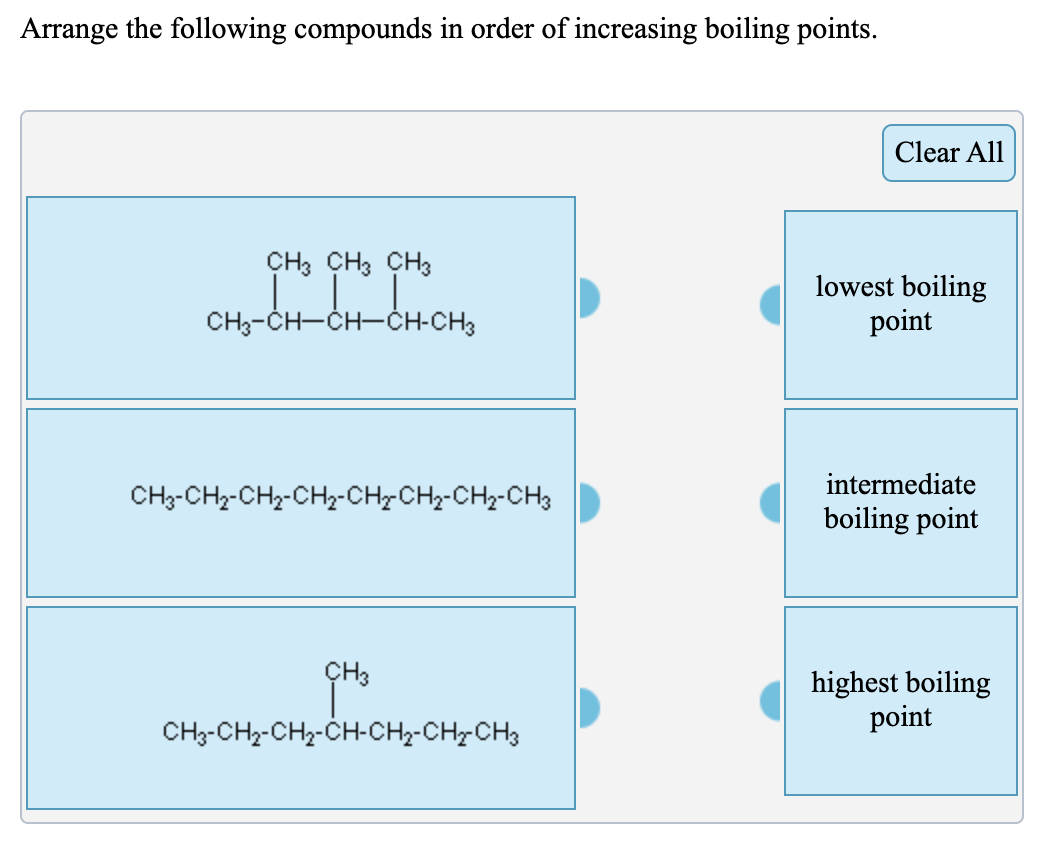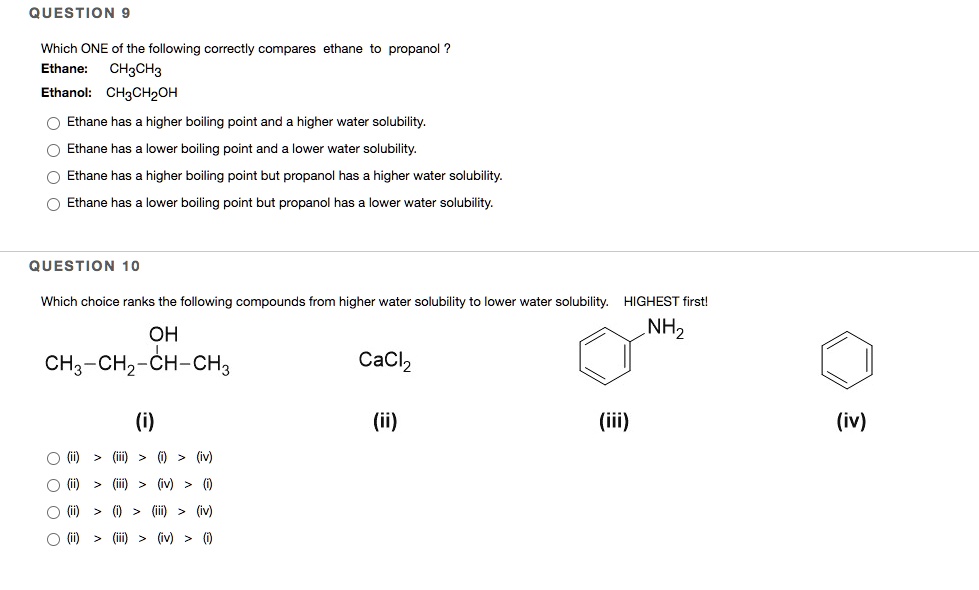
SOLVED: QUESTION Which ONE of the following correctly compares ethane Ethane: CH3CH3 Ethanol: CH:CHzOH propano Ethane has higher boiling point and higher water solubility: Ethane has lower boiling point and lower water

SOLVED:Pick the compound with the highest boiling point in each pair. Explain your reasoning. a. CH3OH or CH3SH b. CH3OCH3 or CH3CH2OH c. CH4 or CH3CH3
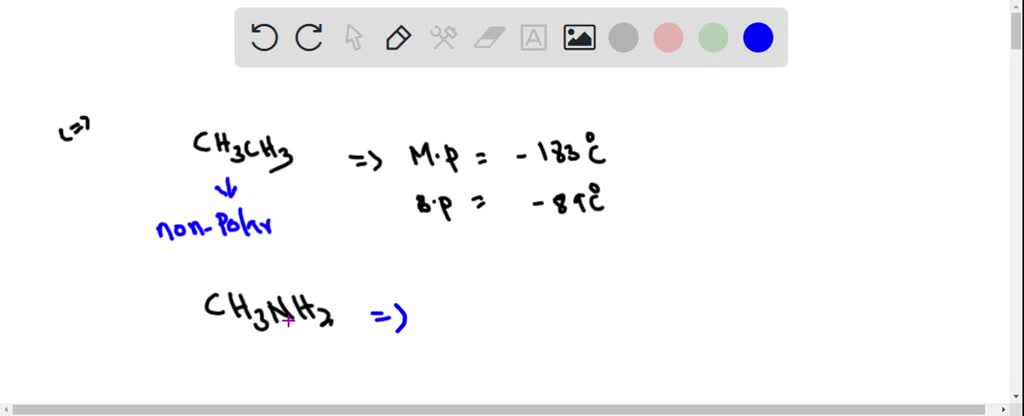
SOLVED: Ethane (CH3CH3) has a melting point of -183°Cand a boiling point of -89°C. Predict the melting point and boiling points for methylamine(CH3NH2)