
Why is the boiling point of diethyl ether lower than that of n-pentane? The structures and molecular weights are similar and ether contains a polar atom. - Quora

Arrange the following compounds in increasing order of their boiling pointsCH3CHO,CH3CH2OH, CH3OCH3, - Brainly.in

Arrange the following compounds in increasing order of their boiling points .CH3CHO,CH3CH2OH,CH3OCH3,CH3CH2CH3
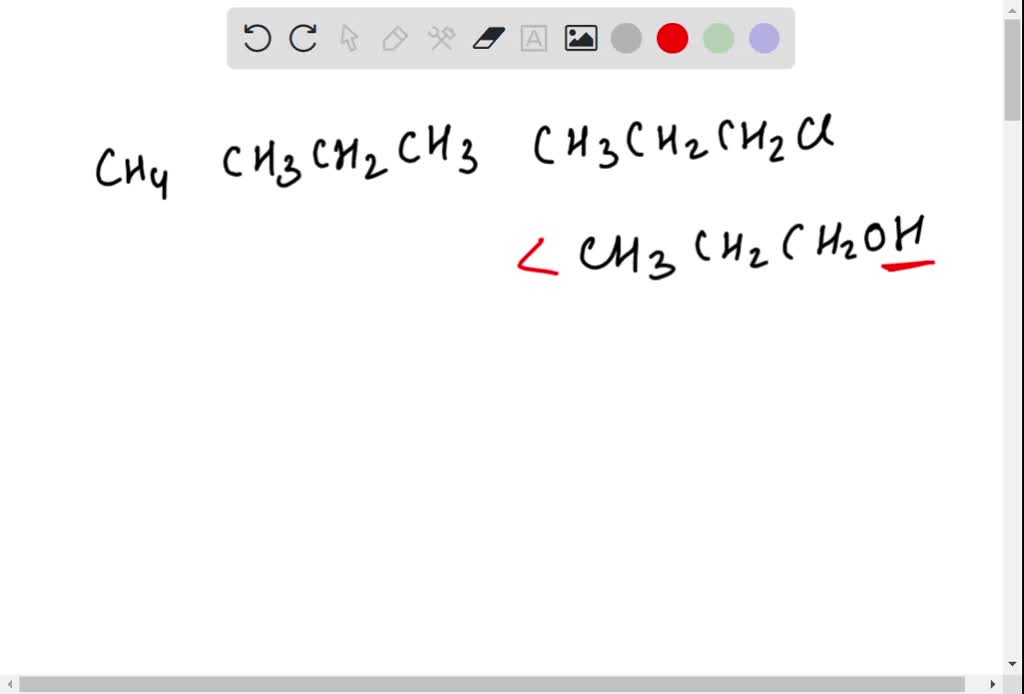
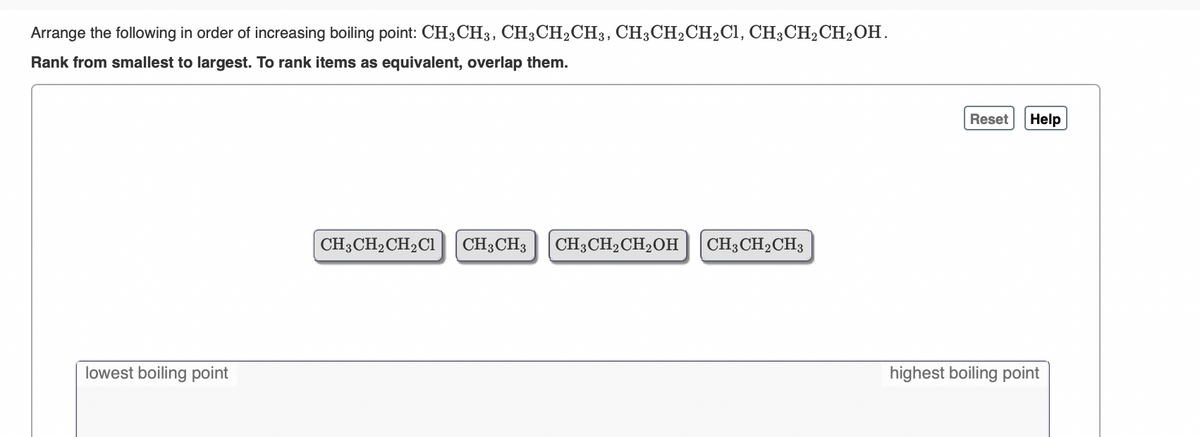
SOLVED: Arrange the following in order of increasing boiling point:CH4 , CH3CH2CH3 , CH3CH2CH2Cl , CH3CH2CH2OH Rank from lowest to highest. To rank items as equivalent, overlap them.
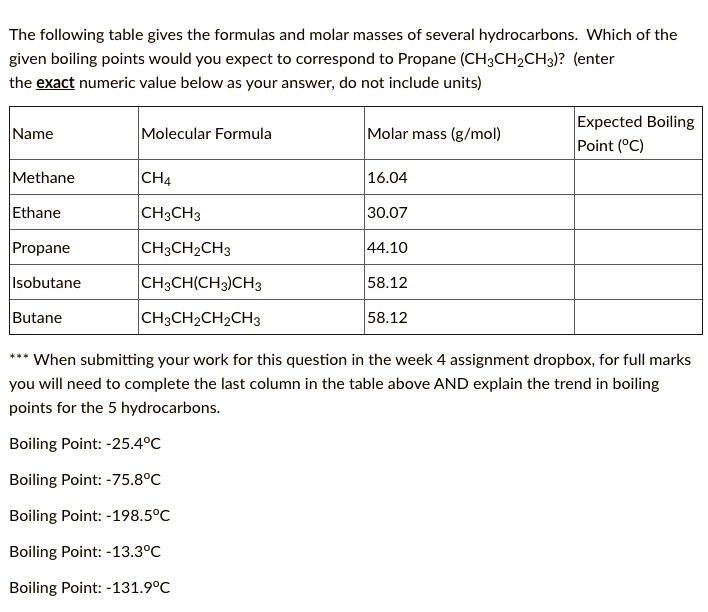
SOLVED: The following table gives the formulas and molar masses of several hydrocarbons: Which of the given boiling points would you expect to correspond to Propane (CH3CH2CH3)? (enter the exact numeric value

Arrange the following compounds in increasing order of their boiling points. CH3CHO, CH3CH2OH, CH3OCH3,CH3CH2CH3

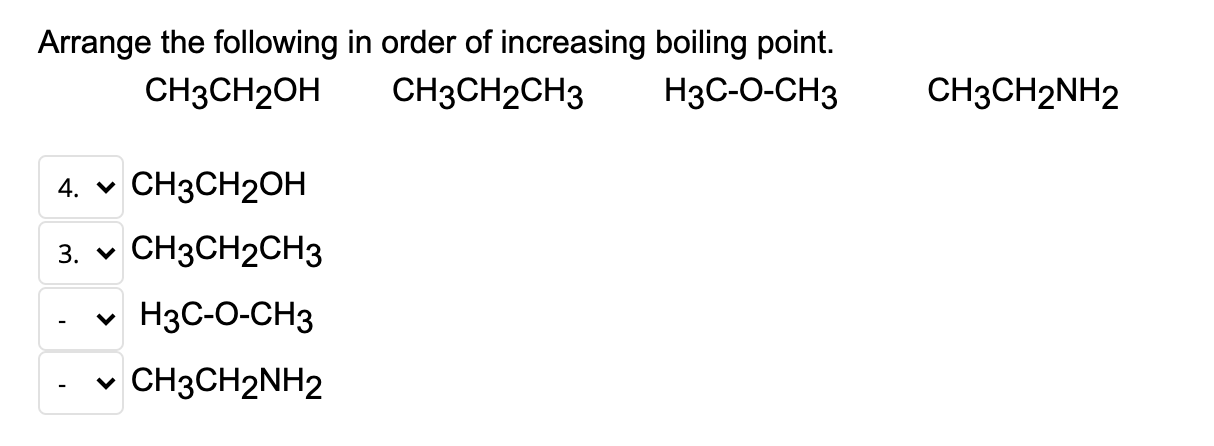
Arrange these compounds in order of increasing boiling point (values in degC are 242, 224, 78, and 118). CH3CH2OH; CH3OCH3; CH3CH2CH3 ; CH3COOH | Homework.Study.com




![Solved] Identify the compound with the highest bo | SolutionInn Solved] Identify the compound with the highest bo | SolutionInn](https://s3.amazonaws.com/si.experts.images/2022/12/63ad594b84a5b_Screenshot20221229143357.png)




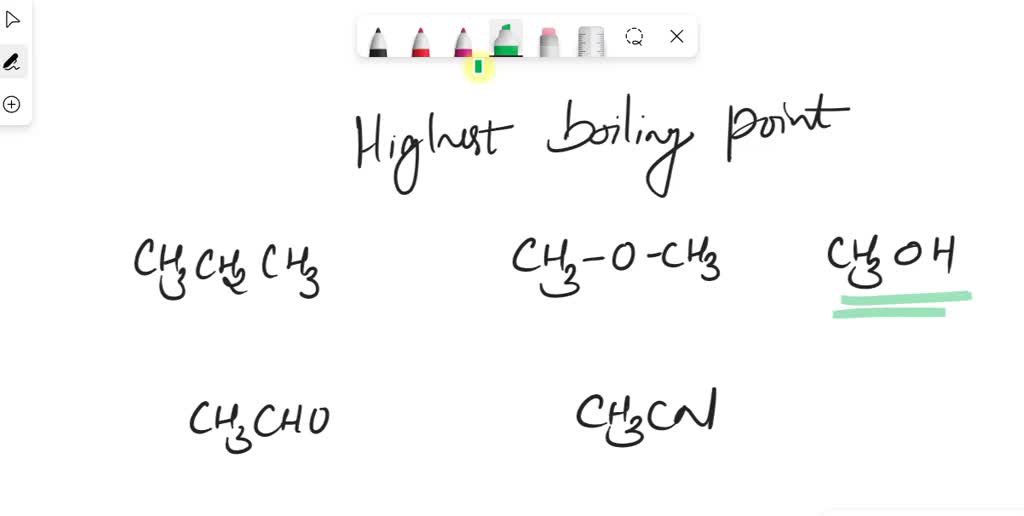
![ANSWERED] 6.The intermolecular force(s) responsible... - Physical Chemistry ANSWERED] 6.The intermolecular force(s) responsible... - Physical Chemistry](https://media.kunduz.com/media/sug-question/raw/45441105-1658671953.6668243.jpeg)

