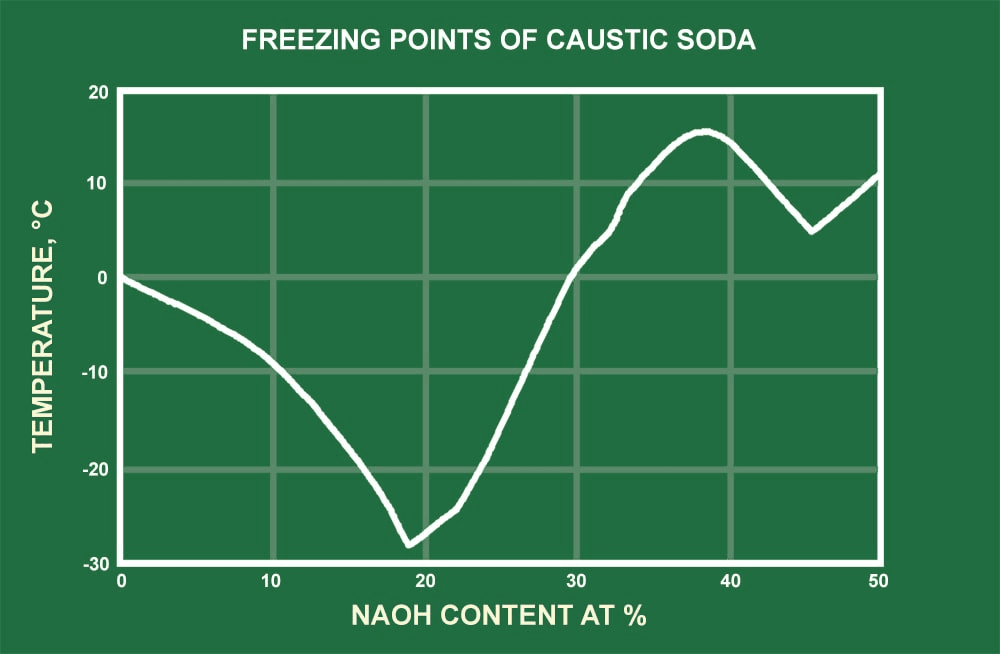
Chemical potentials of solute for aqueous solutions of NaOH and NaCl at... | Download Scientific Diagram
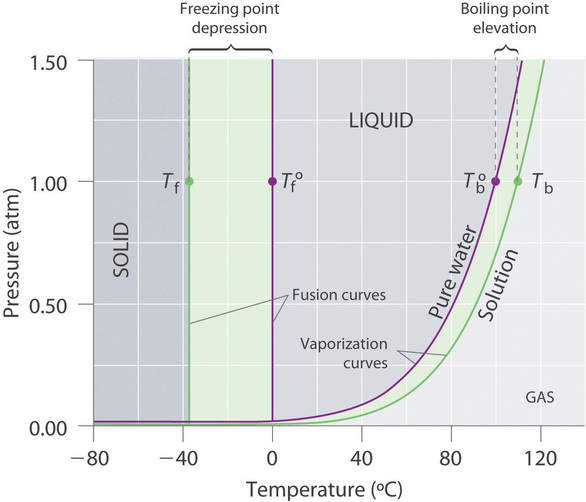
13.8: Freezing-Point Depression and Boiling-Point Elevation of Nonelectrolyte Solutions - Chemistry LibreTexts
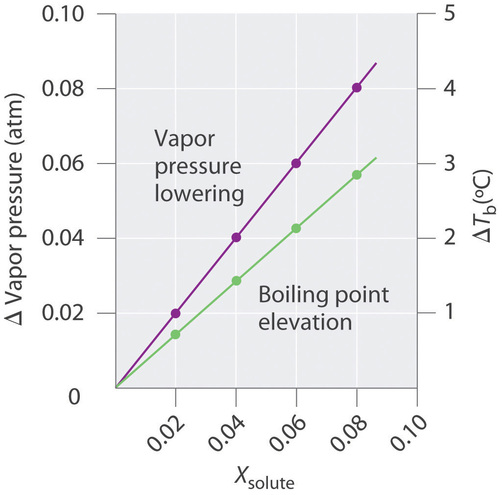
13.8: Freezing-Point Depression and Boiling-Point Elevation of Nonelectrolyte Solutions - Chemistry LibreTexts

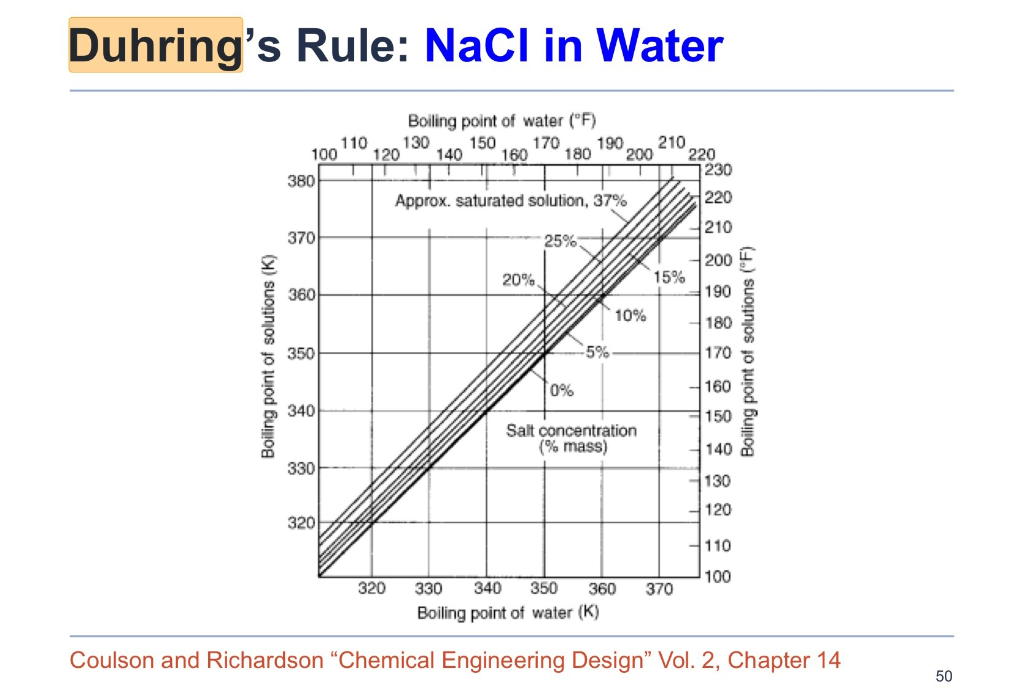
Boiling Point Rise Calculations in Sodium Salt Solutions | Industrial & Engineering Chemistry Research

Boiling temperature at atmospheric pressure of aqueous solutions of... | Download Scientific Diagram
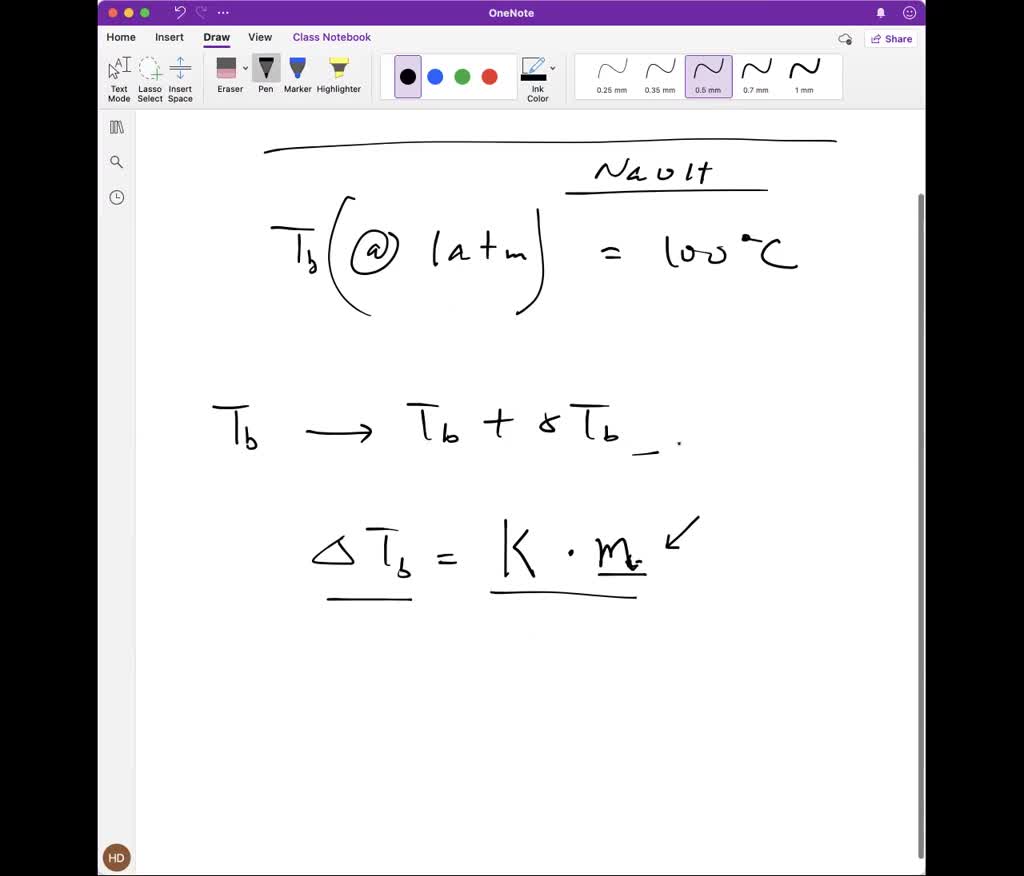
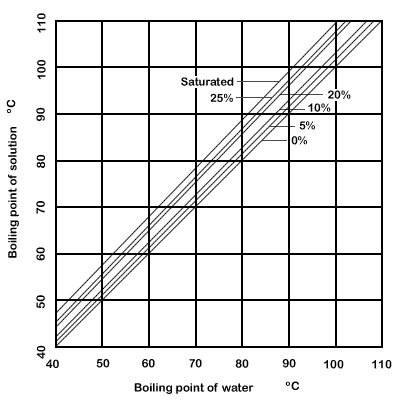
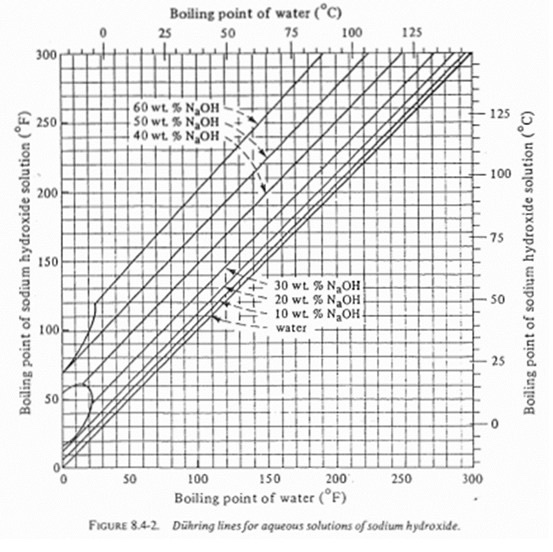
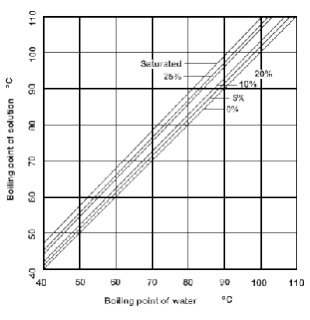
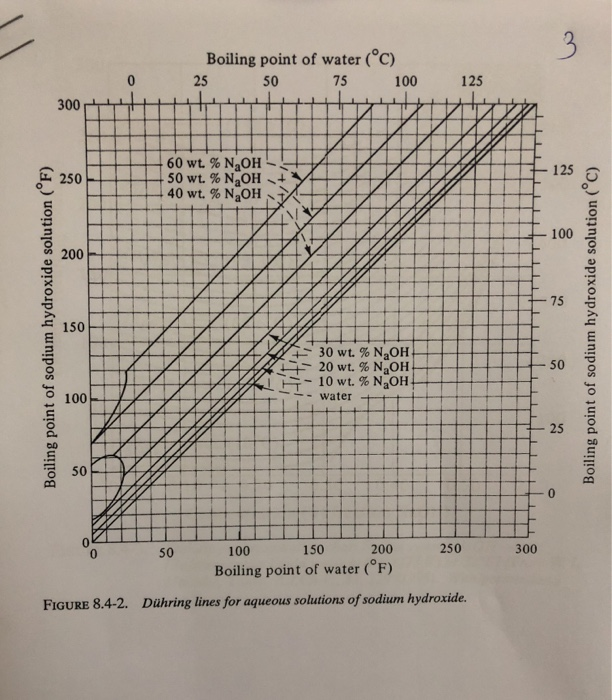
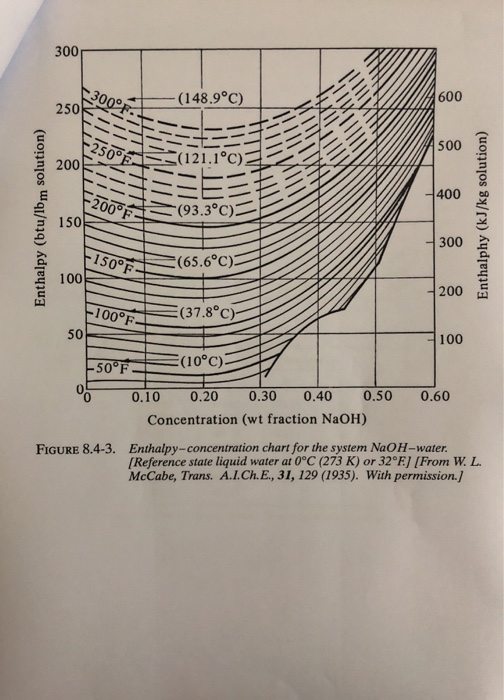
SOLVED: 8.4-5. Determine the boiling temperature of the solution and the boiling-point rise for the following cases. (a) A 30% NaOH solution boiling in an evaporator at a pressure of 172.4 kPa (

Selection of stainless steels for handling sodium hydroxide (NaOH) – British Stainless Steel Association