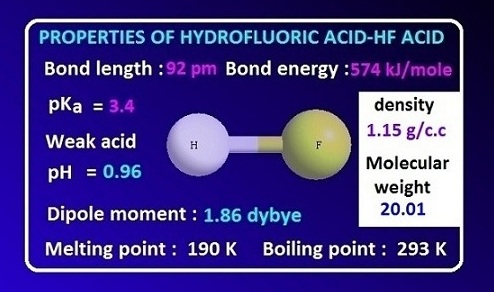
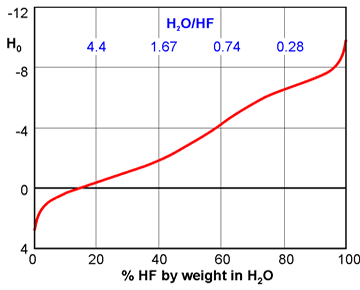
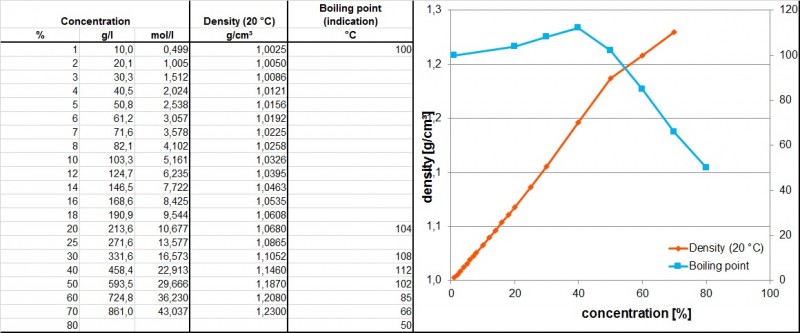
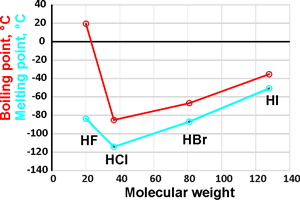
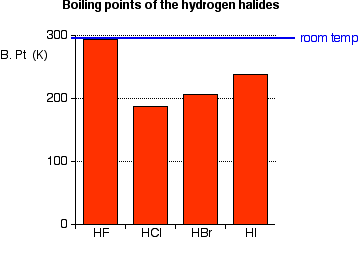
Heat Capacity of Hydrofluoric Acid and (Hydrofluoric Acid + 2-Methylpropane) by Temperature-Pulse Calorimetry | Journal of Chemical & Engineering Data
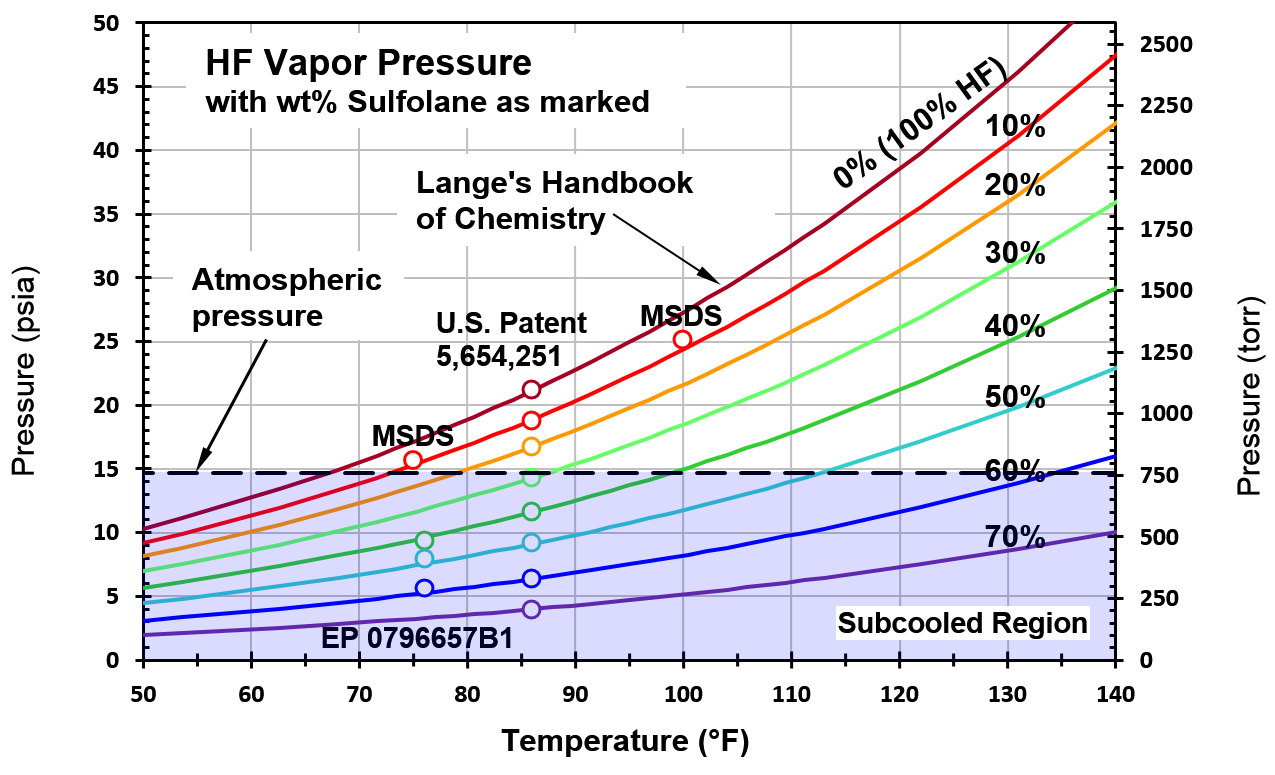
Superheated MHF Excluded from the Only Large-Scale Test Series” by Jim Eninger, Ph.D. – Torrance Refinery Action Alliance

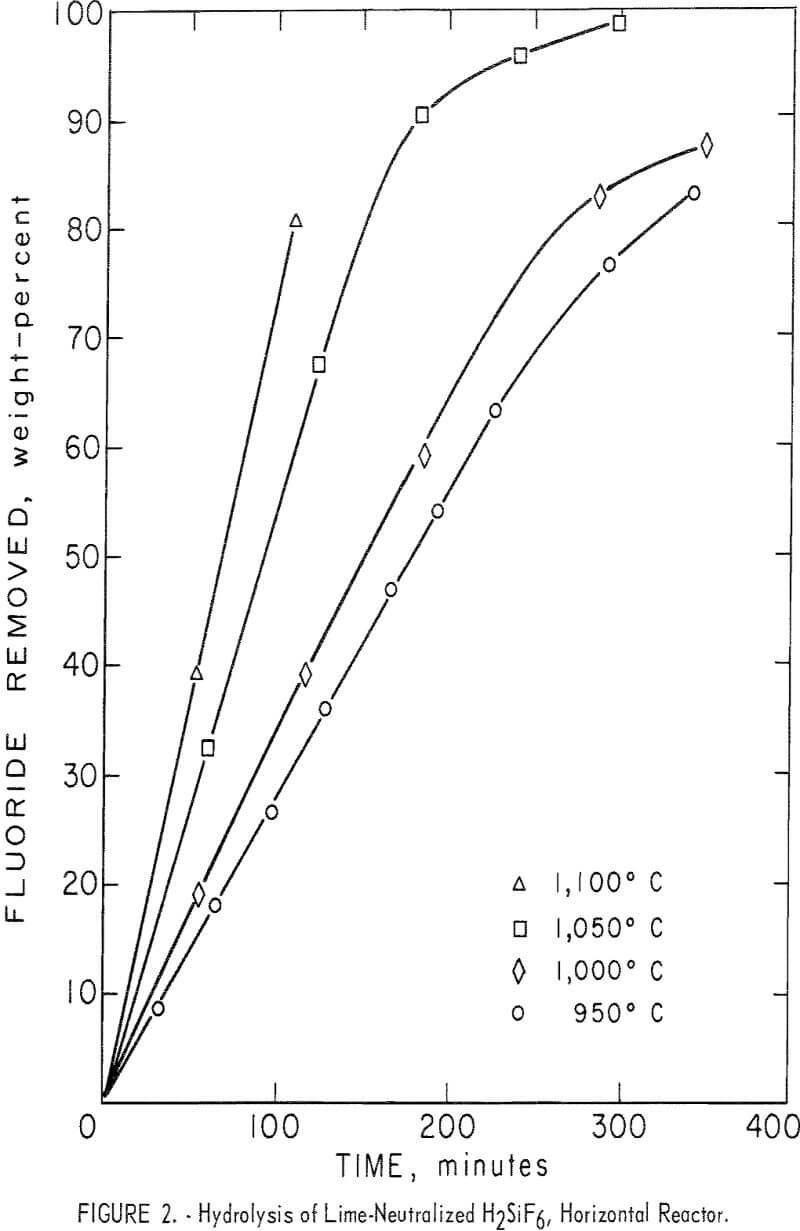
Recycling hydrofluoric acid in the nuclear industry: The OverAzeotreopic Flash process (OVAF) - ScienceDirect

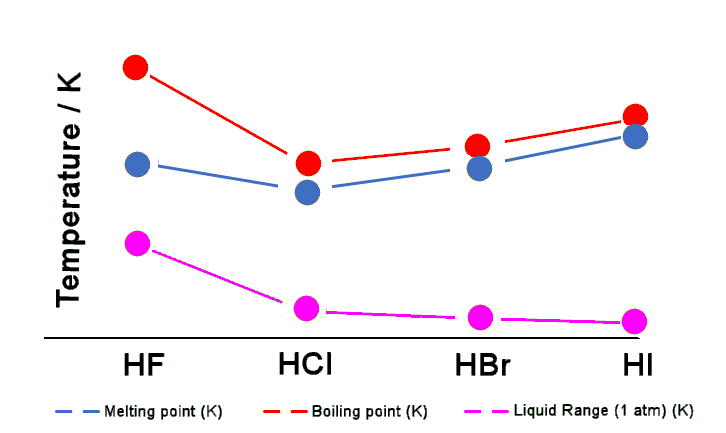
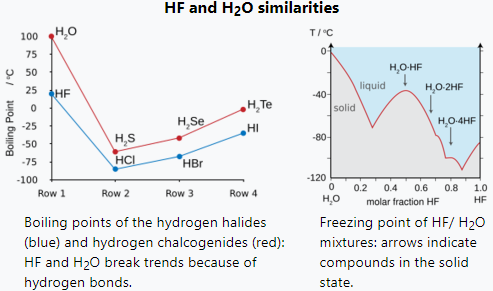
Why does water has a higher boiling point than HF | Hydrogen bonding in water and hydrogen fluoride - YouTube

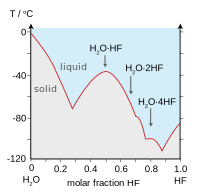
SOLVED: Consider mixture of HF (normal boiling point 19PC and water hydrofluoric acid. Check all statements that apply to it: Select one or more: Hydrofluoric acid forms high-boiling azeotrope The mixture shows