Formation and reshuffling of disulfide bonds in bovine serum albumin demonstrated using tandem mass spectrometry with collision-induced and electron-transfer dissociation | Scientific Reports
Revisiting the conformational state of albumin conjugated to gold nanoclusters: A self-assembly pathway to giant superstructures unraveled | PLOS ONE

Adsorption and Interaction of Bovine Serum Albumin and Pluronic P103 Triblock Copolymer on a Gold Electrode: Double-Layer Capacitance Measurements | ACS Omega
![Illustrative scheme of the bovine serum albumin-BSA (adapted from [39])... | Download Scientific Diagram Illustrative scheme of the bovine serum albumin-BSA (adapted from [39])... | Download Scientific Diagram](https://www.researchgate.net/publication/333009610/figure/fig3/AS:757250243174401@1557553935903/llustrative-scheme-of-the-bovine-serum-albumin-BSA-adapted-from-39-with-the.png)
Illustrative scheme of the bovine serum albumin-BSA (adapted from [39])... | Download Scientific Diagram

Amino Acid Analysis of Pure Protein Hydrolysates with Application of Waters UPLC Amino Acid Analysis Application | Waters
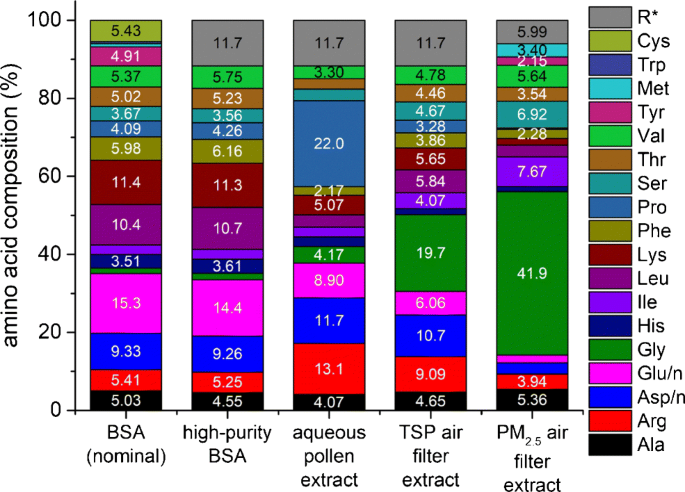
Determination of the protein content of complex samples by aromatic amino acid analysis, liquid chromatography-UV absorbance, and colorimetry | SpringerLink

Materials | Free Full-Text | Conductance Changes in Bovine Serum Albumin Caused by Drug-Binding Triggered Structural Transitions
![PDF] Bovine serum albumin and insulin-dependent diabetes mellitus; is cow's milk still a possible toxicological causative agent of diabetes? | Semantic Scholar PDF] Bovine serum albumin and insulin-dependent diabetes mellitus; is cow's milk still a possible toxicological causative agent of diabetes? | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/92879d23265811d54474f7c414095fe9e9d20a8a/3-Figure2-1.png)
PDF] Bovine serum albumin and insulin-dependent diabetes mellitus; is cow's milk still a possible toxicological causative agent of diabetes? | Semantic Scholar
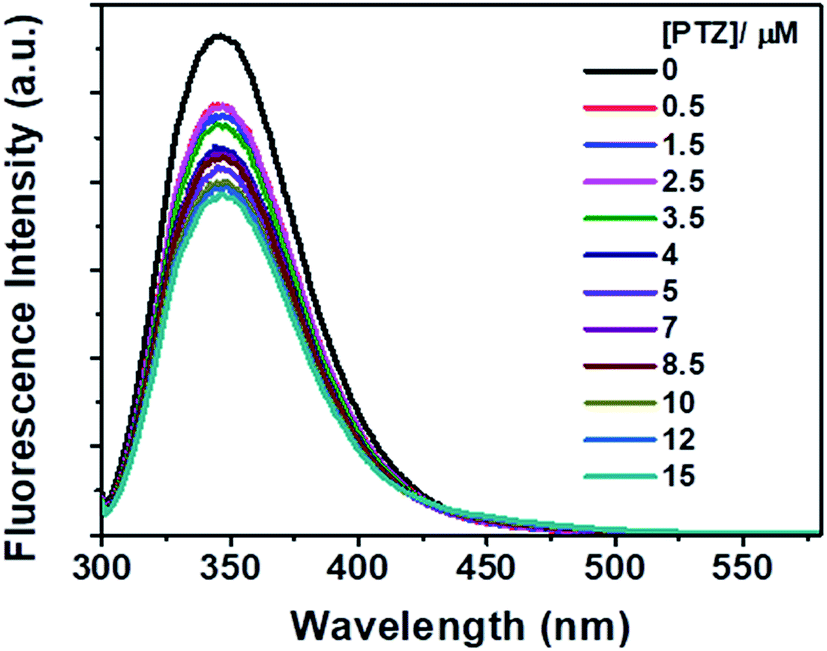
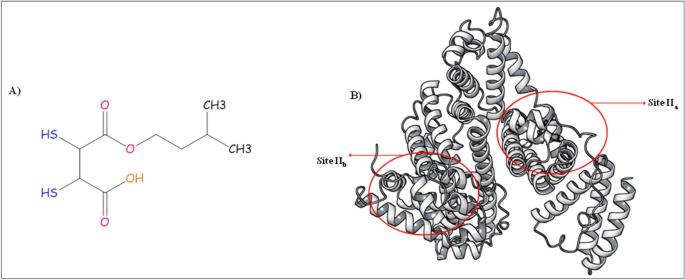
Binding interactions and FRET between bovine serum albumin and various phenothiazine-/anthracene-based dyes: a structure–property relationship - RSC Advances (RSC Publishing) DOI:10.1039/D0RA09580J

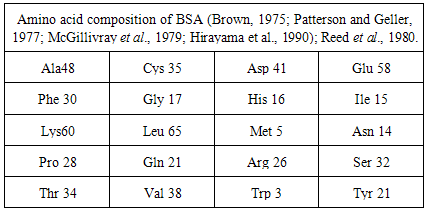
What is the ratio of amino acids in protein bovine serum albumin from 583 amino acids? | ResearchGate

Fewer Exposed Lysine Residues May Explain Relative Resistance of Chicken Serum Albumin to In Vitro Protein Glycation in Comparison to Bovine Serum Albumin | SpringerLink

Figure 3 from The Amino Acid Sequence and Copper ( II )-binding Properties of Peptide ( l-24 ) of Bovine Serum Albumin * | Semantic Scholar

Enzymatic Properties of a Novel Liquefying α-Amylase from an Alkaliphilic Bacillus Isolate and Entire Nucleotide and Amino Acid Sequences | Applied and Environmental Microbiology
Covalently Bound Bovine Serum Albumin (BSA) Protein Modified Hydrogenated Diamond Like Carbon (HDLC) Surface as Biosensor Application

Exploring how structural and dynamic properties of bovine and canine serum albumins differ from human serum albumin - ScienceDirect

Probing three-dimensional structure of bovine serum albumin by chemical cross-linking and mass spectrometry - ScienceDirect