Quantitative Analysis of the Substrate Specificity of Human Rhinovirus 3C Protease and Exploration of Its Substrate Recognition Mechanisms | ACS Chemical Biology

SARS-CoV 3CLpro cleavage sites and the canonical recognition sequence.... | Download Scientific Diagram
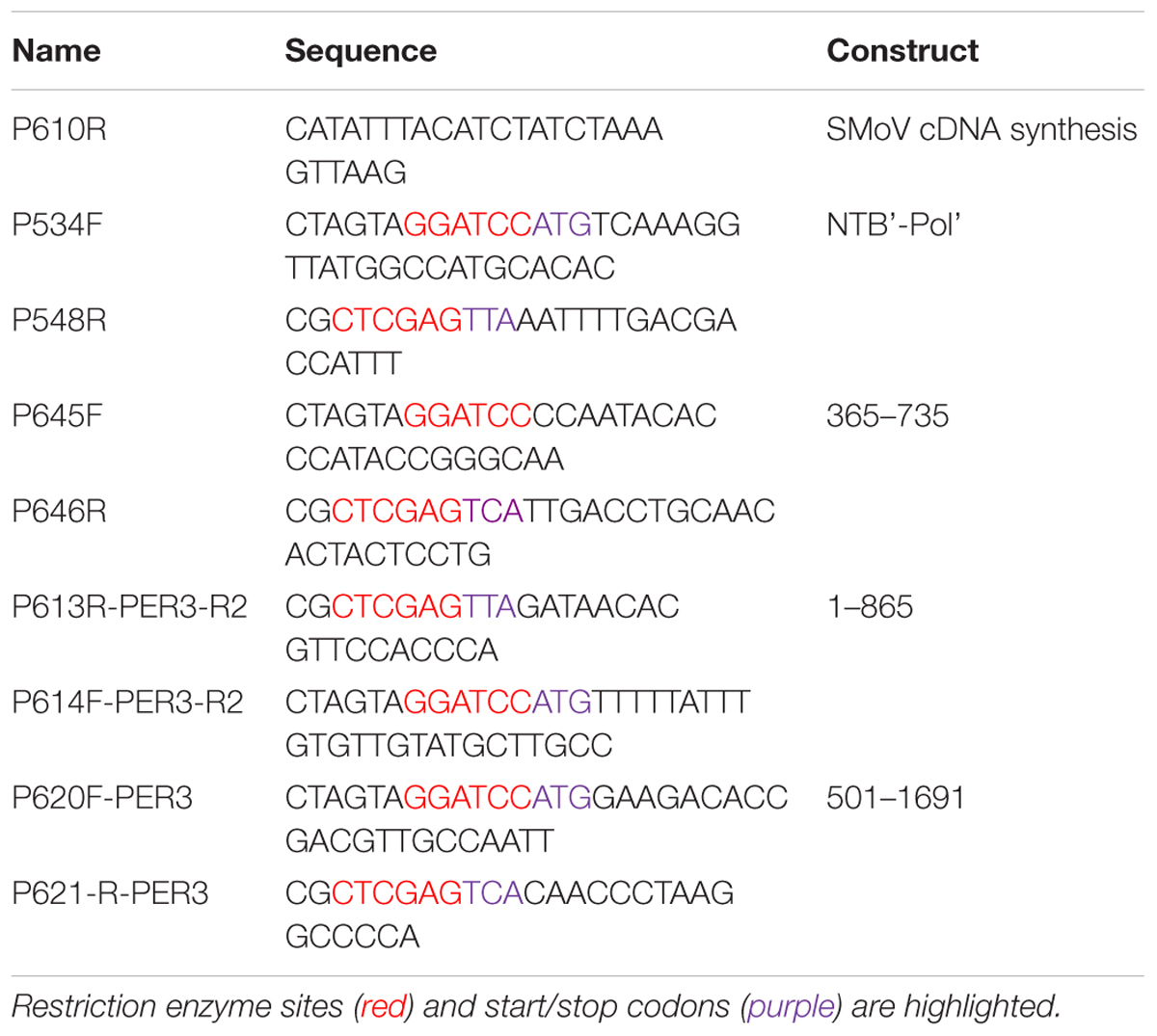
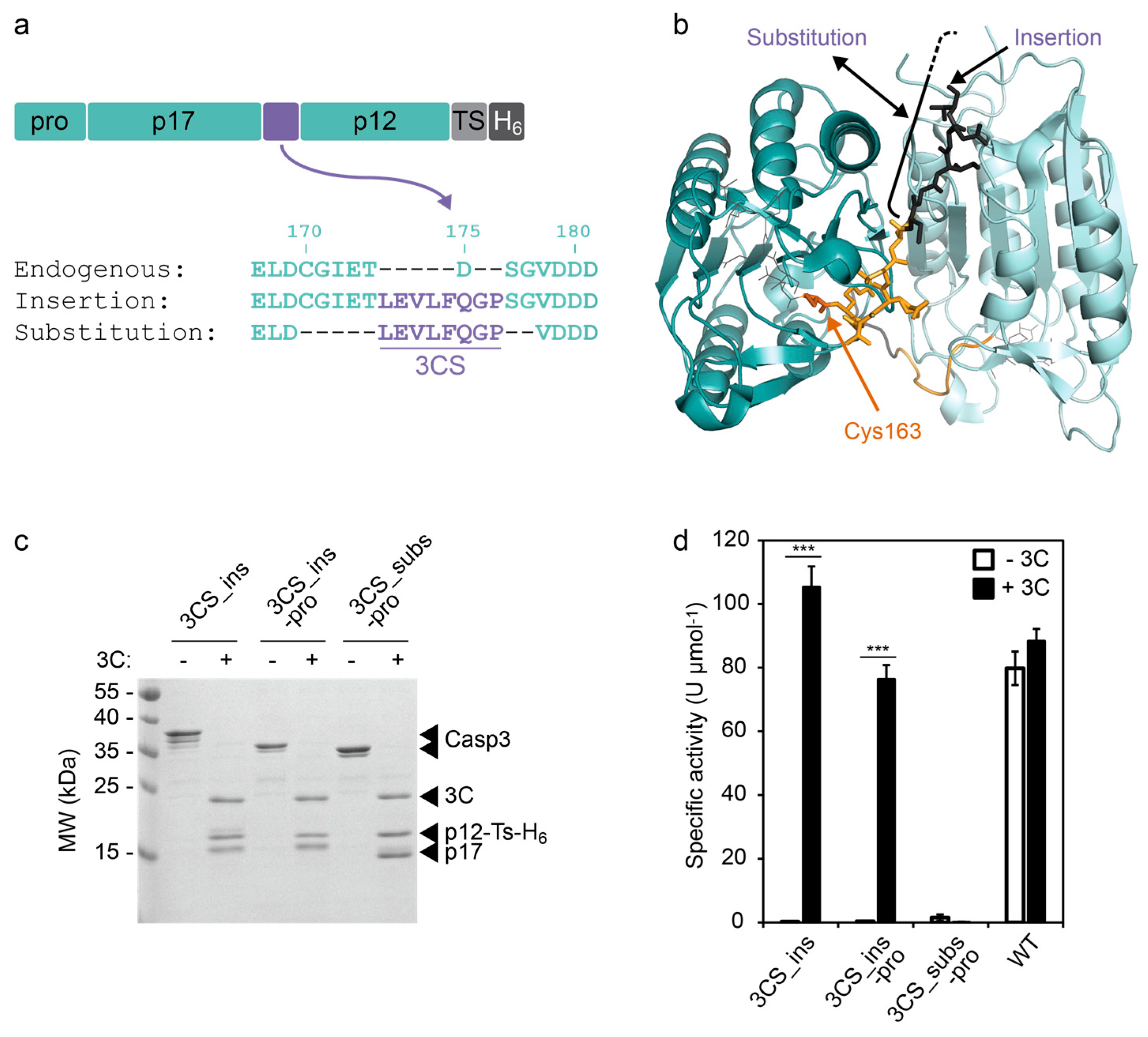
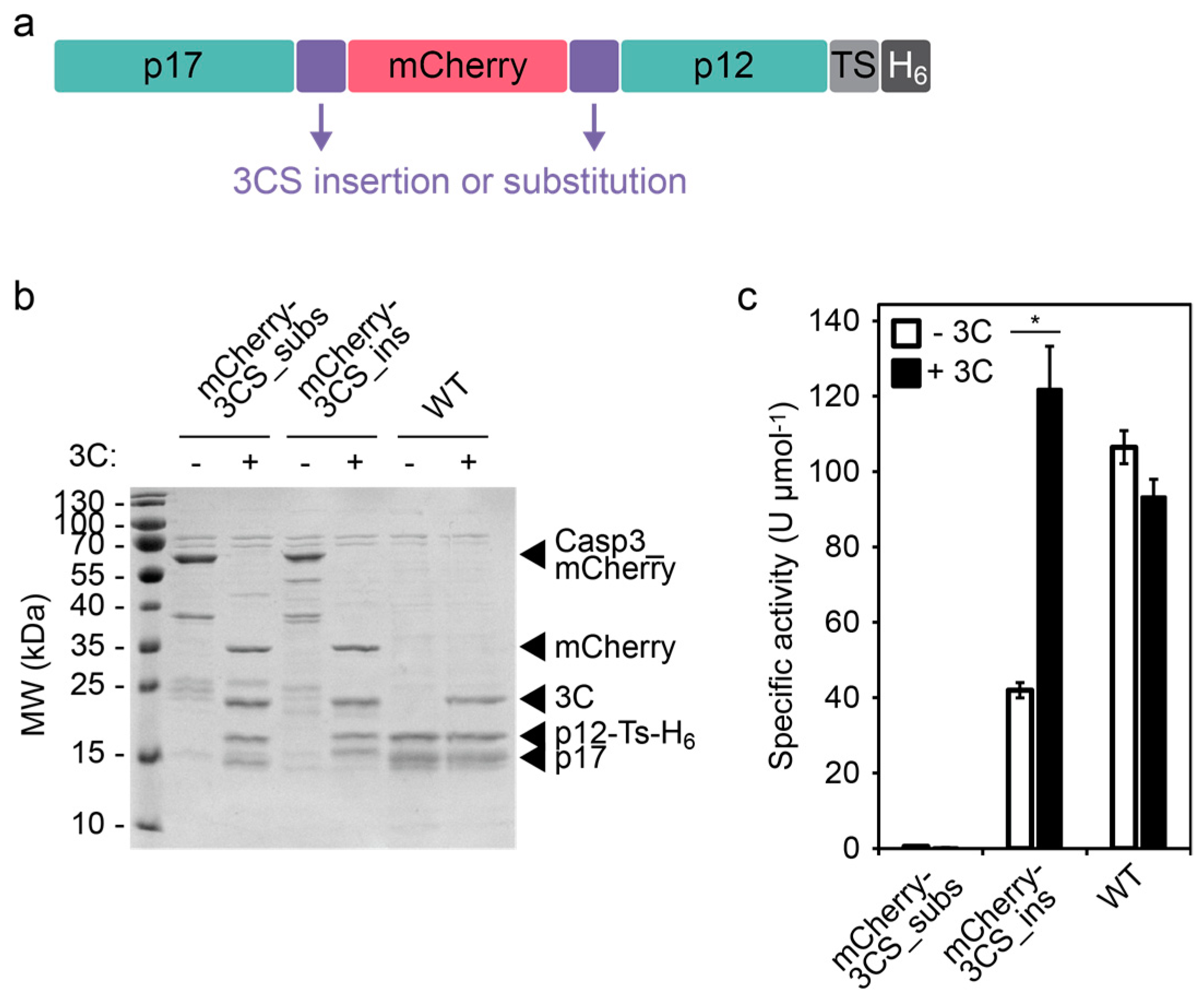
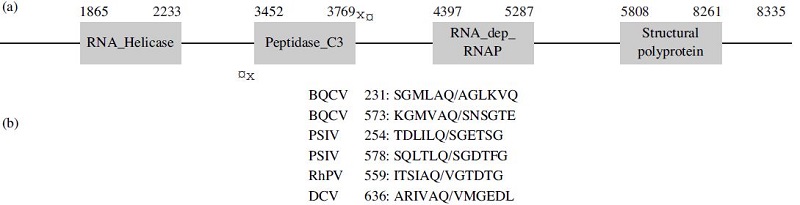
Frontiers | Identification of Cleavage Sites Recognized by the 3C-Like Cysteine Protease within the Two Polyproteins of Strawberry Mottle Virus

A split protease-E. coli ClpXP system quantifies protein–protein interactions in Escherichia coli cells | Communications Biology

Frontiers | Identification of Cleavage Sites Recognized by the 3C-Like Cysteine Protease within the Two Polyproteins of Strawberry Mottle Virus

SDS-PAGE showing the HRV 3C protease activity in the presence various... | Download Scientific Diagram

A) Three tags, i.e. 10x-His, FLAG and HA, and one proteolytic site for... | Download Scientific Diagram

NT*-HRV3CP: An optimized construct of human rhinovirus 14 3C protease for high-yield expression and fast affinity-tag cleavage - ScienceDirect

A simple solid phase, peptide-based fluorescent assay for the efficient and universal screening of HRV 3C protease inhibitors - ScienceDirect